DRY FASTING CONCLUSION
March 1, 2016
Aframomum Melegueta
October 4, 2017The Fasting Phoenix: A Dance of Renewal
In the realm where thirst and hunger dwell,
A tale of trial and magic I shall tell.
Of days when sustenance is but a dream,
And life, a dry and desolate stream.
A hopeful sprite in marrow’s deep abyss,
Clings to mirages of a water’s kiss.
Yet, finds not but the cruel, empty air,
And echoes of a banquet that’s not there.
The specter, cloaked in shadows of the end,
To mortal coil a message, it does send.
But in the silence of the void and dearth,
A key is found, a magic of rebirth.
From what gear does this mechanism run?
From survival’s wheel, beneath the fasting sun.
In the crucible of the body’s plight,
The fittest of the cells ignite.
A purge of weakness, a cleansing fire,
From the depths, a phoenix does aspire.
No feeder of disease can find its keep,
In this fortress where the strong do sleep.
The non-essentials, in their vanity,
Are reduced to mere inanity.
Recycled, transmuted in the void,
To sustain that which cannot be destroyed.
A deep renewal, a cellular ballet,
Unattainable by any other way.
In the stark silence of the fasting dawn,
A new creation, from the old, is drawn.
So, in the riddle of the fasting night,
Find the magic in the body’s flight.
For in deprivation, stark and bare,
Lie the secrets of the life we dare.
22/2, 44/4, 66/6, 88/8: INTERMITTENT DRY FASTING WITH SUPERTONIC SCIENCE BASED ANTI-AGING HERBS FOR SUPREME HEALTH AND LONGEVITY
DRY FASTING: THE ULTIMATE PATH TO LONGEVITY — A TRUE LIFE OR DEATH SCENARIO
On extended dry fasts the first few days are the roughest as the body is still optimistically holding out for food or water but once it figures out that water and food are not coming and that death is imminent if it doesn’t adapt and fast to given circumstance it switches gears into survival mode— this highly adapted state IS the magical key of dry fasting. Survival of the fittest. All weakness is eradicated. The option of supporting the diseased and parasitic is no longer viable. All non essentials are eliminated and recycled to sustain the essentials. This IS cellular renewal at the deepest level— unattainable by any other means.
A 5 DAY DRY FAST SCIENTIFIC STUDY:
“Background: Although there is considerable research in the field of fasting and fluid restriction, little is known about the impact of food and water deprivation (FWD) on body circumferences and vital parameters.
Methods: During 5 days of FWD in 10 healthy adults, hemodynamic, metabolic, and renal parameters, such as weight, 5 circumferences at neck, waist, hip, chest at axilla, chest at nipples, and 1 new oblique hip circumference were measured daily. For each circumference, new quotients of daily circumference-to-weight decrease were calculated. The set of employed parameters quantified and monitored dieting persons’ compliance and efficacy of the method.
Results: The values of blood pressure, heart rate, hemoglobin oxygen saturation, glucose, K+, Na+, Cl–, urea, creatinine, and serum osmolality proved to be stable. The mean creatinine clearance increased up to 167%. The mean daily weight decrease (1,390 ± 60 g) demonstrated the effectiveness of FWD in weight reduction. The daily decrease of all measured circumferences and the values of the corresponding circumference-to-weight decrease quotients reflected considerable volume decrease in all measured body parts per day and kg of weight loss during FWD.
Conclusion: The intervention of 5 FWD days in 10 healthy adults was found to be safe, decreased weight and all measured circumferences, and improved renal function considerably.”
http://www.karger.com/Article/Abstract/357718
While indeed longer duration dry fasts, 3 to 7 days and beyond, are best utilized to bring an ill or obese person back into balance—once homeostasis is restored daily dry (or zero calorie fluid restricted) fasting is the most effective disease preventive measure one can employ in maintaining optimal health and delaying aging.
Looking for the SECRET BLENDS?
10,000 Scientific Studies on Fasting, Calorie Restriction & Extending Life Span (CLICK HERE)
IMPORTANT: The exact reason why fluid is TEMPORARILY restricted will be explained in depth below; ***the goal is NOT chronic dehydration***as that would obviously have negative consequences; downregulation of Klotho being one.
WHAT IS KLOTHO? Klotho, a transmembrane protein, protease, and hormone mainly expressed in renal tissue counteracts aging. Overexpression of Klotho substantially prolongs the life span. Klotho deficiency leads to excessive formation of 1,25(OH)2D3, growth deficit, accelerated aging, and early death. Aging is frequently paralleled by dehydration, which is considered to accelerate the development of age-related disorders.
The present study explored the possibility that dehydration influences Klotho expression. Klotho transcript levels were determined by RT-PCR, and Klotho protein abundance was detected by Western blotting in renal tissue from hydrated and 36-h-dehydrated mice as well as in human embryonic kidney (HEK293) cells.
Dehydration was followed by a significant decline of renal Klotho transcript levels and protein abundance, accompanied by an increase in plasma osmolarity as well as plasma ADH, aldosterone, and 1,25(OH)2D3 levels. Antidiuretic hormone (ADH; 50 nM) and aldosterone (1 μM) significantly decreased Klotho transcription and protein expression in HEK293 cells. In conclusion, the present observations disclose a powerful effect of dehydration on Klotho expression, an effect at least partially mediated by enhanced release of ADH and aldosterone.
http://www.physiology.org/doi/full/10.1152/ajprenal.00037.2011
This is the how and why behind dry fasting.
DAILY DRY FASTING in conjunction with anti-aging herbs and diet— the SCIENCE behind it.
For daily dry fasting which is your wisest protocol for maintaining a precision tuned body built for the long haul a 1-6 hour eating window is recommended with your highest level of activity including workout around this time as well.
In other words:
23:1, 22:2, 21:3, 20:4, 19:5 or 18:6
1st number dry (or at the very least fluid restricted), 2nd number feeding/hydration window.
RESTRICTION OF FEEDING TIME —WHY?
**It’s not what you eat so much as WHEN you eat. **
“Feeding schedule has been shown to have a signifiant impact on health and survival. In studies, time-restricted feeding had profound effects on neural, peripheral, and cardiovascular physiology and improved sleep, body weight maintenance, and delayed signs of cardiac aging, under UNCHANGED caloric intake and activity.”
http://www.ncbi.nlm.nih.gov/…/…/pdf/f1000research-5-7686.pdf
THE PILLARS OF LONGEVITY
There are 3 factors we are focusing on that promotes longevity and absence of disease:
AMPK ACTIVATION
mTOR INHIBITION
AUTOPHAGY
More here: https://scholar.google.com/scholar
All 3 are activated by KETOSIS* which is a rapid result of DRY FASTING. (No water or food.)
*KETONE BODIES STIMULATE CHAPERONE-MEDIATED AUTOPHAGY
“Interestingly, the increase in the concentration of circulating ketone bodies parallels the induction of CHAPERONE-MEDIATED AUTOPHAGY (CMA), which is also activated by prolonged starvation.
In these studies we have demonstrated that ketone bodies, more specifically BOH, stimulate CMA by causing the oxidation of substrates. In addition, during prolonged starvation CMA is activated because of increased lamp2a in the lysosomal membrane and increased lyhsc70 in the lysosomal lumen (9, 31). Our data indicate that ketone bodies can also stimulate CMA by affecting substrate proteins during prolonged starvation in vivo. This finding gives us further insight into the physiological importance of CMA stimulation during times of nutrient deprivation.”
http://m.jbc.org/content/280/27/25864.full
Chaperone-mediated autophagy: roles in disease and aging
http://www.nature.com/cr/journal/v24/n1/pdf/cr2013153a.pdf
LYSOSOMES
To understand autophagy you need to understand lysosomes. Watch this brief video explaining what they are.
http://study.com/…/l…/lysosome-definition-function-quiz.html
EXCELLENT VIDEO visually explaining the 3 types of AUTOPHAGY:
1. Macroautophagy
2. Micrautophagy
3. Chaperone-Mediated Autophagy
WATCH HERE: https://m.youtube.com/watch?v=BiwnJtYCuww
Skip the fake ketones and burn more fat
“Our results demonstrate that during fasting, circulating ketone bodies exert an inhibitory influence on the rate of ketogenesis. This mechanism might play an important role in preventing the development of uncontrolled hyperketonemia during starvation.”
That means if you are eating exogenous ketones and fat you are dramatically inhibiting the production of ketones from your own body fat. NO BUENO.
https://www.sciencedirect.com/science/article/pii/002604957590092X
WHY NO WATER?
The ketogenic diet (KD) is traditionally introduced with an initial period of fasting and fluid restriction. *Since the 1930s it has been known that fluid restricted fasting accelerates ketosis which in turn has positive effects on preventing epilepsy.
http://digitalcommons.unmc.edu/cgi/viewcontent.cgi…
IMPORTANT: Over-hydrate 1-2 gallons of water a day for 3-7 days to downregulate aldosterone before beginning. Once you pull back on the water on your dry fast the fat will fall right off you.
https://scholar.google.com/scholar
Question: If dehydration RAISES blood sugar and fasting LOWERS blood sugar —what happens when you combine them together as in dry fasting?
ULTRA RAPID ONSET OF KETOSIS!
“Fasting resulted in reduced plasma glucose concentrations compared with the control study, while dehydration resulted in increased plasma glucose concentrations compared with the control study (P < .001). Glucose production and disposal were decreased during the fasting study and increased during the dehydration study compared with the control study.
***Glucagon concentrations and rates of development of ketosis and metabolic acidosis were increased during both fasting and dehydration compared with control.***
These data suggest that fasting and dehydration have differential effects on glycemia during insulin deficiency, with dehydration favoring the development of hyperglycemia and fasting resulting in reduced glucose concentrations.”
http://www.ncbi.nlm.nih.gov/m/pubmed/11229425/
Here’s the numbers:
Dry fasting = Ketosis in 12-24 hours
Water fasting = Ketosis in 48-72 hours.
Test it and see for yourself.
Get ketostix: http://www.theketogenicdiet.org/ketostix/
What is Glucagon?
Glucagon is a peptide hormone, produced by alpha cells of the pancreas. It works to raise the concentration of glucose in the bloodstream. Its effect is opposite that of insulin, which lowers the glucose.
Why is KETOSIS important and what is it’s link to autophagy, mTOR inhibition, AMPK activation?
https://scholar.google.com/scholar
Calorie restriction and its effects on aging:
https://scholar.google.com/scholar
All about mTOR: http://selfhacked.com/…/11/03/mtor-natural-mtor-inhibitors/
DRY FASTING (DEHYDRATION) INHIBITS MTOR
Direct inactivation of mTOR, stimulation of the AMP-activated protein kinase, and the destabilization of individual proteins may impair mTOR signaling under dehydrating conditions.
http://onlinelibrary.wiley.com/doi/10.1111/j.1748-1716.2006.01547.x/full
What Is AMPK?
“AMPK stands for adenosine monophosphate-activated protein kinase. It is found in every living cell of every living mammal (and most other animals) on Earth. If you want to avoid the life span-shortening symptoms of aging, you need to maintain optimal AMPK activity.
AMPK has been referred to as a “metabolic master switch.” AMPK controls a gamut of metabolic pathways that enable us to extract energy from food, store and distribute that energy safely through the body, and ultimately use that energy for everything from moving and mating to talking and thinking, and even to understanding these very words as you read them.
The core role of AMPK is to sense each cell’s energy status at every moment, and to trigger responses that maintain the cell’s energy at precisely the optimum level. Too little available energy starves the cell, while too much energy can exhaust and disrupt cellular components.In either case (too little or too much energy), the cell (and the tissues, organs, and systems in which it is a part) functions inefficiently. That energy inefficiency ultimately leads to the dysfunctions we identify as the diseases (or symptoms) of aging.
Here’s how AMPK works: Every cell in your body depends absolutely on a steady supply of energy in the form of chemical bonds. When you eat and absorb nutrients, energy from chemical bonds in food is released and passed down a complex series of enzymes until it is stored again in a molecule called adenosine triphosphate, or ATP. The more ATP that is present in the cell, the higher the cell’s available energy supply. When ATP is broken down to release energy for cellular work, a major end product is adenosine monophosphate, or AMP.
In preclinical research, enhanced AMPK activity has been associated with a 20-30% increase in life span, but that’s just the beginning of the health benefits conferred by this critical cellular enzyme.
Increased AMPK activation has been shown to help reduce fat storage (especially dangerous belly fat), increase insulin sensitivity (to lower blood glucose), reducecholesterol/triglyceride production, and suppress chronic inflammation. All of these factors underlie the lethal diseases of aging.”
SCIATICA PAIN? ACTIVATE AMPK
“Hence, AMPK activators suppress translation regulation pathways in sensory neurons and inhibit nascent protein synthesis in the sciatic nerve associated with PNI (peripheral nerve injury).”
https://molecularpain.biomedcentral.com/articles/10.1186/1744-8069-7-70
CHRONIC PAIN? ACTIVATE AMPK & INHIBIT MTOR
“AMPK: An emerging target for modification of injury-induced pain plasticity
Chronic pain is a critical medical problem afflicting hundreds of millions of people worldwide with costly effects on society and health care systems. Novel therapeutic avenues for the treatment of pain are needed that are directly targeted to the molecular mechanisms that promote and maintain chronic pain states. Recent evidence suggests that peripheral pain plasticity is promoted and potentially maintained via changes in translation control that are mediated by mTORC1 and MAPK pathways. While these pathways can be targeted individually, stimulating the AMPK pathway with direct or indirect activators achieves inhibition of these pathways via engagement of a single kinase. Here we review the form, function and pharmacology of AMPK with special attention to its emerging role as a potential target for pain therapeutics. We present the existing evidence supporting a role of AMPK activation in alleviating symptoms of peripheral nerve injury- and incision-induced pain plasticity and the blockade of the development of chronic pain following surgery. We argue that these preclinical findings support a strong rationale for clinical trials of currently available AMPK activators and further development of novel pharmacological strategies for more potent and efficacious manipulation of AMPK in the clinical setting. Finally, we posit that AMPK represents a unique opportunity for drug development in the kinase area for pain because it is pharmacologically manipulated via activation rather than inhibition potentially offering a wider therapeutic window with interesting additional pharmacological opportunities. Altogether, the physiology, pharmacology and therapeutic opportunities surrounding AMPK make it an attractive target for novel intervention for chronic pain and its prevention.” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3844111/
ALCOHOL, mTOR & LONGEVITY
Here is the scientific connection on how alcohol inhibits mTOR and promotes longevity:
“Many studies have suggested that moderate alcohol consumption reduces mortality. There is also substantial evidence that lifespan is extended with suppression of TOR (target of rapamycin). It was reported recently that rapamycin is able to extend the lifespan of a mammal—implicating the mammalian TOR (mTOR). mTOR has a requirement for the lipid second messenger phosphatidic acid (PA), which is generated by phospholipase D (PLD). Therefore, in principle, suppression of PLD would be similar to treatment with rapamycin. Significantly, PLD utilizes ethanol preferentially over water in the hydrolysis reaction that ordinarily generates PA. In the presence of ethanol, phosphatidyl-ethanol is generated at the expense of PA leading to the suppression of mTOR. This reaction, known as the transphosphatidylation reaction, provides a mechanistic basis for the reduced mortality observed with moderate consumption of alcohol—that being the suppression of mTOR.”
USE HIGH HEAT SAUNA DAILY TO ACTIVATE AMPK AND INCREASE INSULIN SENSITIVITY THUS PREVENTING DIABETES AND OBESITY
“Heat shock protein inducers share metabolic pathways associated with exercise with activation of AMPK, PGC1-a, and sirtuins. Diabetic therapies that induce the stress response, whether via heat, bioactive compounds, or genetic manipulation, improve or prevent all of the morbidities and comorbidities associated with the disease. The agents reduce insulin resistance, inflammatory cytokines, visceral adiposity, and body weight while increasing mitochondrial activity, normalizing membrane structure and lipid composition, and preserving organ function. Therapies restoring the stress response can re-tip the balance from disease into health and address the multifaceted defects associated with the disease.
One striking observation is how a brief heat shock—as short as 15 min and as infrequent as once a week—results in remarkable improvements in the metabolic state. A variety of techniques have been used to induce hyperthermia—hot water immersion, warm electric blanket, sauna, and infrared box. Mild, direct electrical current stimulation has also been used to augment heat-induced rises in HSPs. Relevantly, whole-body hyperthermia raises baseline iHSPs (Shinohara et al. 2006; Singleton and Wischmeyer 2006).”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4041942/
Autophagy is the best Way to Get Rid of (Cellular) Junk.
“Autophagy is the way your cells “clean house” and “recycle the trash”. Along with the ubiquitin proteasome system, autophagy is one of the main methods that cells use to clear dysfunctional or misfolded proteins. Autophagy can clear any kind of trash: intracellular viruses, bacteria, damaged proteins, protein aggregates and subcellular organelles.” http://www.anti-agingfirewalls.com/…/autophagy-the-houseke
How AUTOPHAGY regulates AGING:
“The accumulation of cellular damage is a feature common to all aging cells and leads to decreased ability of the organism to survive. The overall rate at which damage accumulates is influenced by conserved metabolic factors (longevity pathways and regulatory proteins) that control lifespan through adjusting mechanisms for maintenance and repair. Autophagy, the major catabolic process of eukaryotic cells that degrades and recycles damaged macromolecules and organelles, is implicated in aging and in the incidence of diverse age-related pathologies. Recent evidence has revealed that autophagic activity is required for lifespan extension in various long-lived mutant organisms, and that numerous autophagy-related genes or proteins are directly regulated by longevity pathways. These findings support the emerging view that autophagy is a central regulatory mechanism for aging in diverse eukaryotic species.” https://scholar.google.com/scholar
AMPK and autophagy are ALSO activated by intense exercise
“Activation of autophagy in human skeletal muscle is dependent on exercise intensity and AMPK activation
In summary, the present study is to our knowledge the first to provide evidence that exercise-induced autophagy relies on the activation of the AMPK pathway in human skeletal muscle. In addition, our findings indicate that the most effective strategy to activate autophagy depends on exercise intensity rather than dietary supply, thus providing interesting clues to improve the understanding of remodeling in both healthy and pathologic human skeletal muscle.” http://www.fasebj.org/doi/full/10.1096/fj.14-267187
Exercise ENHANCES rather than inhibits mTOR and increases protein synthesis (muscle growth) so you want to time it near feeding window.
“In summary, the current investigation provides additional evidence that endurance exercise does not compromise the anabolic stimulus provided by subsequent strength training. Our combined training enhanced the expression of both Akt and mTOR protein, as well as the areas of type I and type II fibers, whereas strength training only led to elevated protein content of mTOR and increased size of type II fibers. In addition, correlations between changes in the levels of Akt and mTOR protein and changes in type I area and the mean fiber area indicate that these proteins play an important role in hypertrophy.” http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0149082
OVERWHELMING EVIDENCE: Aging controlled by mTOR
“There is overwhelming evidence that cellular mechanisms and signaling pathways regulating ageing are controlled by mTOR. Here we have highlighted important studies that support a role for both mTOR dependent protein synthesis and autophagy in ageing. Genetic approaches show a clear overlap between signaling networks that control ageing and autophagy. An increase in autophagy controlled by mTOR extends life span via dietary restriction and insulin signaling. ” http://citeseerx.ist.psu.edu/viewdoc/download…
The LESS you keep the MTOR pathway open or active the GREATER your health will be and the LONGER you will live.
Growth or longevity. One or the other.
Here’s your percentages of mTOR activation based on feeding and intense exercise window times:
6 hour window is 25%
5 hour window is 20%,
4 hour window is 16%,
3 hour window is 12%,
2 hour window is 8%,
1 hour window is 4%.
The smaller the window the better.
HIGH INTENSITY INTERVAL (HIIT) EXERCISE
The type of exercise I recommend is of short duration and peak intensity. No lolly gagging! Hour or less. Legs one day; upper body next. Too much exercise is not good and prematurely wears the body out. Intense in and out. Cardio ideas: Sprints are wonderful. Swimming laps are great as well as stationary bike intervals 10 seconds (peak exertion) LEVEL 10 followed by 50 seconds (rest) LEVEL 1 for 15-20 cycles.
⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐
“I NEED EXACT INSTRUCTIONS ON HOW TO LOSE 30 POUNDS IN 21 DAYS.”
⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐
No problem, here ya go!
FIRST THING IN MORNING:
1/4-1/2 tsp Seven Sages
1/8 tsp Thermo
1/8 tsp Trinity
1/4 tsp Matcha
1/4 tsp 20:1 Spice (1/8 200:1)
1/4 tsp 20:1 Peel (1/8 200:1)
In 4-6 oz COFFEE
repeat again afternoon if needed or skip if not needed.
PRE WORKOUT:
1/8 tsp Thermo
1/8-1/4 tsp Nebula (BEAST MODE)
1/4 tsp Matcha
in shot of espresso.
1 hour cardio.
Dinner 7 pm
SALAD:
Arugula 2 cups
Parsley 1 cup
Spice 1 tsp (1/4 tsp 200:1)
Peel 1 tsp (1/4 tsp 200:1)
Shilajit 1/4 tsp (Fulvic acid/minerals)
Pine pollen 1 TBS
1 oz olive oil
1/4 cup lemon or lime juice
1-2 oz Braggs Vinaigrette (optional)
Salt and pepper to taste
TOTAL DAILY LIQUID INTAKE: 16oz
Add electrolyte tablets to water as needed if low energy.
1/8-1/4 tsp Trinity before bed for sleep.
Repeat.
Do this and NOTHING ELSE you will lose 1.5 lbs on average a day until all fat GONE.
EASY!
Get these 2 combos:
#1 https://theinterstellarplan.com/product/super-ultimate-sampler-pack-sevensages/
#2 https://theinterstellarplan.com/product/spiceandpeelcombo/
Use discount code DRYFAST to save 10%.
22/2: intermittent (fluid restricted) fasting – A Great place to start
(cliffnote version / immediate action plan/ what I have done for years)
Here’s the LIFE CHANGING 22/2 intermittent fasting protocol:
💥MORNING
Cognitive enhancement
Trinity 1/4 tsp
Autonomous 1/8 tsp (very strong start small)
Seven Sages (sold separately) 1/4 tsp (if you feel good increase to 1/2 tsp)
Rewire 1/8-1/4 tsp
Matcha 1/4 tsp (too much at once may make you feel nauseous; very strong matcha)
Jing Force 1/4 tsp
Glucose Blocker 1/8 tsp
Anti aging (must be while fasting low insulin for maximum effects)
Senolytic 1/4 tsp
ACB 1/4 tsp
Autophagy 1/4 tsp
In cold brew or hot coffee
💥MIDDAY Pre workout
Energy, Strength, Power
1/4 tsp Nebula
1/4 tsp Thermo
1/4 tsp Anti Adipogenic
1/8 tsp Glucose Blocker
1/4 tsp Matcha
In cold brew or hot coffee
💥FEEDING WINDOW
Hair / Skin / Immunity
Super Tonic Hair 1/4 tsp
Pine pollen 1 tbs
Peel 1/4 tsp
Spice 1/4 tsp
Beauty Blend 1/4 tsp
Victorious 1/4 tsp
Shilajit 1/2 tsp
Luteolin 1/4 tsp
Glucose Blocker 1/8 tsp
In fresh grapefruit juice
1/8-1/4 tsp PURGE with low insulin index paleo keto meal.
💥BEDTIME
Quality sleep
1/4 tsp Hypnotic
1/8 tsp Glucose Blocker
In warm chamomile tea
💥22/2 means 22 hour fast with coffee and blends / 2 hour paleo keto low insulin index meal daily (fresh green salad with olive oil vinaigrette is perfect) I prefer Mediterranean Diet (see below)
💵 BEST VALUE 👉 https://theinterstellarplan.com/product/monthlyspecial/
💥Samples are also available here:
https://theinterstellarplan.com/product/200to1samples/
💥Why intermittent fasting is important: https://theinterstellarplan.com/2021/02/intermittent-fasting/
💥Time restricted feeding benefits:
https://theinterstellarplan.com/2021/12/time-restricted-feeding/
💥MTOR: the rapid aging pathway and how and why you want to inhibit it:
https://theinterstellarplan.com/2019/04/mtor-articles/
💥Autophagy and it’s importance:
https://theinterstellarplan.com/2019/04/autophagy/
💥Oxidative stress:
https://theinterstellarplan.com/2019/04/autophagy/
💥Mediterranean Diet: https://theinterstellarplan.com/2022/02/mediterranean-diet/
💥Blends are best taken in coffee while fasting here’s why: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5611980/ https://pubmed.ncbi.nlm.nih.gov/28526373/
💥If weight loss is goal get this: https://theinterstellarplan.com/product/challengecombo/
Why OLIVE OIL IS exceptional and should be consumed daily
Extra virgin olive oil polyphenols, especially oleuropeinand oleocanthal, downregulate the insulin/IGF1/AKT /mTOR pathway which thus activates FOXO3 favoring longevitywith anti-cancer, anti-diabetic, anti-inflammatoryand neuroprotectiveeffects.
INTERSTELLaR LONGEVITY SHAKE for 2 HOUR FEEDING WINDOW
(click links to read benefits of each)
2-3 tbs pine pollen, blueberries (pterostilbene) , goji berries, Brazil nuts (selenium), 1 tsp vitamin E oil, collagen, Matcha, Trinity, Seven Sages, Shilajit, about 4 oz of fresh squeezed OJ (the rest water), 1-2 tbs cacao, 1 oz extra virgin olive oil, 1/8 tsp each: cinnamon, cloves, nutmeg, allspice, ginger, sage, rosemary, thyme, turmeric,caraway, cumin, fennel, cardamom, peppermint, star anise, orange peel. (Or keep it simple and get Interstellar Spice and Peel.) That’s over 250 longevity ingredients combined!
Talk about FEELING AMAZING!!!!
44/4
(THE NEXT LEVEL)
Advanced intermittent dry fasting:
Once you practice with 22/2 for a while you are ready to take it to the next level which is 44 hour dry fast (air only if you like or 1 Oz shot espresso every 4-6 hours with 1/8 tsp of your favorite blends) with a 4 hour feast and rehydration session. This is my personal favorite but I alternate between 22/2, 44/4 and 66/6 (ultra advanced) depending on what I got going on. If you want mega results fast do 44/4 or 66/6, it’s more challenging and requires more mental discipline but the results are worth it. Simply repeat the 44/4 or 66/6 cycle until all fat and and all health problems are GONE.
*Incorporate Interstellar Longevity Spice shake (with ACB) during 4 hour window as with 22/2
To kick it up a notch further combine 44/4 with ice baths to mega release FGF21 which has incredible health effects on the body and shown to dramatically increase lifespan in mice by 40%.
Note: Cold exposure activates mTOR so you definitely want to make sure you are inhibiting mTOR with ACB during the 4 hour feeding/hydration window.
I have an ice bath in my garage which I keep at 40 degrees year round. I will sit in it for 10-20 minutes one to three times a day. Any time you feel hungry, thirsty or fatigued an ice bath will make you feel incredible again and you will forget you are even fasting. In the winter if cold enough I will go jump in the pool and swim laps for same effect.
66/6
(ultra advanced level)
For the 66/6 (master 44/4 first) I recommend 1 Oz shot of espresso and 1/8 tsp blends every 4 hours (so 4 oz total liquid intake a day) during the 66 and then a 6 hour feast/rehydration session. * Be sure to drink water until urine is clear during the 6. REPEAT.
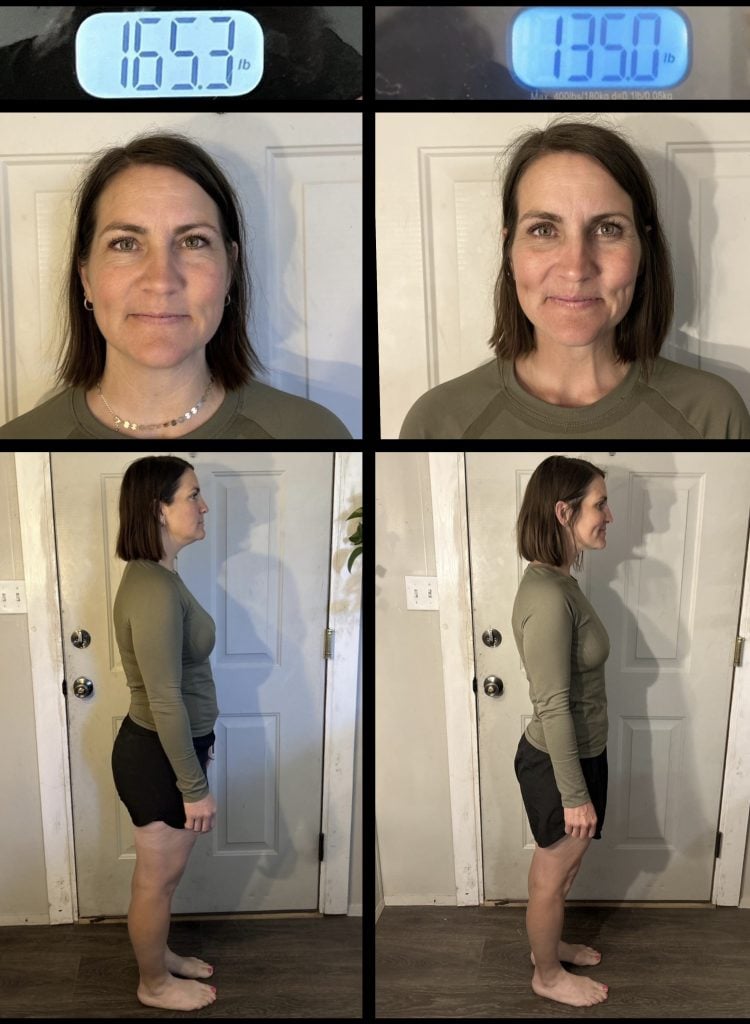
*If you average 4-7 lbs net loss weighing in at the 66 hour mark each cycle that’s 40-70 lbs lost in 30 days!!!!!
?$750 prize in store credit if you lose 50lbs in 30 days and post pix to group documenting it with time stamped photos. Must take photos every 66 hour weigh in: face, front, side, back, scale, blood sugar, blood pressure, ketosix. (See pinned post in group for instructions or message me.)
?$250 prize in store credit for 30lbs in 30 days.
?$500 prize In store credit for 40lbs in 30 days.
88/8
(Jedi level)
This is not really practical or sustainable unless you have 60+ lbs to lose and are EXTREMELY DEDICATED and are willing to stop at nothing.
Same rules as 66/6 except now it’s 4 days (88 hours dry or fluid restructed). Four 1 oz shots of espresso with 1/8 tsp blends are allowed a day.
?$1000 prize in store credit if you post daily pix in group and lose 60lbs in 32 days doing the 88/8 method. You should lose 6-8lbs every 88 weigh in and between 50 and 70lbs in 32 days. (8 cycles of 88/8).
Message me if you want to do it.
Example of how to use the blends on 44/4 66/6 88/8:
Start fast set timer for 4 hours (or when you wake up)
1st 1oz espresso shot:
1/2 tsp Seven Sages (the day)
1/4 tsp Matcha
1/4 tsp 200:1 Peel
1/4 tsp Shilajit
1/8 tsp ACB
(reset timer)
4 hours later
2nd 1oz espresso shot:
1/4 tsp Thermo
1/4 tsp Matcha
1/4 tsp 200:1 Spice
1/8 tsp ACB
(reset timer)
4 hours later
3rd 1oz espresso shot:
1/4 tsp Nebula
1/4 tsp Matcha
1/4 tsp Shilajit
1/4 tsp 200:1 Spice
1/8 tsp ACB
(reset timer)
4 hours later
4th 1oz espresso shot:
1/4 tsp Trinity
1/4 tsp Matcha
1/4 tsp 200:1 Peel
1/8 tsp ACB
Keep Trinity, Thermo, Nebula, Seven Sages in separate shots.
4 x 4 hours covers 16 hours the other 8 hours for sleep = 24 hours. Repeat.
COFFEE INHIBITS MTOR (hence the espresso shots with blends on the challenge every 4 hours)
Coffee consumption increases energy production and decreases hepatic mTOR levels!
-
The present study investigated the locomotor activity, energy metabolism, and lipid metabolism of aged mice fed coffee. Regular and decaffeinated coffee consumption decreased plasma-free fatty acid levels, increased hepatic adenosine triphosphate content, and decreased total mammalian target of rapamycin (mTOR) and phosphorylated mTOR (p-mTOR) protein content in the liver. Consumption of regular coffee increased the food and water intake, locomotor activity, volume of oxygen consumption, and respiration exchange ratio of aged mice. Coffee, one of the world’s most consumed beverages, has potential antiaging effects.
-
“Regular and decaffeinated coffee consumption decreased hepatic total mTOR and p-mTOR levels independently of Akt and AMPK pathways in aged mice. Because decreased mTORC1 activity is known to have antiaging effects, coffee consumption during old age may retard aging. Moreover, coffee consumption by the aged population had a positive effect on behavioral energy and lipid metabolism”
WHICH IS THE BEST BLEND OR COMBO TO START WITH?
⭐️⭐️⭐️⭐️⭐️ If you have never tried the blends before get the SUPER ULTIMATE SAMPLER W/ SEVEN SAGES
This gets you 7 blends to try at a 30% discount which enables you to then identify your favorite(s). I regularly utilize them all.
Usually, I have found, people don’t have just one issue but a bunch combined and have a hard time identifying or pinpointing where to begin; this combo covers all the bases and results are generally noticeable within the first week.
For maximum benefit; I would consider incorporating daily intermittent fasting (see 22/2 section) with the blends to not only restore vibrant health (for those with any health issues) but also dramatically slow down the aging process while also neutralizing oxidative stress (the precursor to most disease) —the blends make the fasting part easy and keep you from binging on junk food and sweets activating insulin and mTOR (the rapid aging pathway).
INSTRUCTIONS for combining SUPER ULTIMATE or *ACB 10 BLEND COMBO (see below) with 22/2 (I do this regularly) intermittent fluid restricted fasting:
Start your day off (breakfast time) with:
1/8 tsp Trinity with
1/2 tsp Seven Sages
1/4 tsp Matcha
*1/8-1/4 tsp ACB, 200:1 PEEL & 200:1 SPICE
in 1-4oz espresso or coffee (the less you drink the faster the fast works)
Midday (lunch time):
1/4 tsp Matcha
1/8 -1/4 tsp Nebula (or Thermo —you can rotate on different days)
*1/8-1/4 tsp ACB, 200:1 PEEL, 200:1 SPICE
In 1-4oz espresso or coffee
And later in feeding window in 8-16oz grapefruit, blueberry, strawberry juice:
1 -3 TBS Pine Pollen
1/2 tsp Shilajit
1/4 tsp Matcha
1/8 -1/4 tsp Thermo (or Nebula)
*1/8-1/4 tsp ACB, 200:1 PEEL, 200:1 SPICE
BONUS: Add 1/2 tsp APIGENIN, THUNDER and/or NIAGRA (bought separately)
Half hour before bed:
1/8 tsp Trinity in hot water.
What you can likely expect after a few weeks:
- No anxiety,
- No irritability,
- No constant gnawing hunger,
- Enhanced mental clarity,
- Elevated mood and outlook on life,
- Depression and melancholy nonexistent,
- Tons of energy,
- Deep, rejuvenating, blissful sleep,
- Aches, pains and inflammation subside,
- Radiant glowing skin and complexion,
- More muscle, strength and endurance,
- Enhanced libido,
- Body fat begins to disappear,
- Blood sugar and blood pressure normalizes,
- Allergies gone or greatly reduced,
- Immune system bolstered, you rarely get sick,
- Renewed inspiration and creativity,
- Basically you are going to be feeling unstoppable, like a BRAND NEW YOU
ATTENTION: Do be prepared — as your friends and family will definitely all be AMAZED with how INCREDIBLE you look and how happy you are and will all be asking, ”What on earth are you doing???” *Hint: Carry business cards around with you (10 included in every order) so you can easily send them all to the website to read up on the SCIENCE behind the blends themselves.
The BEST VALUE (“the GO BIG or GO HOME combo”) is the ACB 10 Blend Combo featuring the super ultimate Sampler w/ 200:1 Peel & Spice:
Back to the science!!!!
INSULIN AND AGING:
YOU WANT TO KEEP YOUR INSULIN LEVELS LOW? AND YOUR INSULIN SENSITIVITY HIGH?
“There is considerable evidence that the IIS signaling pathway is also involved in control of aging and longevity in mammals. A Major increase in average and maximal lifespan in mutant mice with reduced plasma levels of IGF-1 and insulin was reported in 1996,9 and subsequently other mouse mutants with reduced IIS signaling were shown to be long-lived. However, identifying the specific role of insulin and separating it from the effects of reduced GH and IGF-1 signaling in these mutants is very difficult.
The only study published to date that specifically addresses the impact of an isolated primary alteration in insulin signaling on lifespan in mammals is a report of increased longevity of mice with disruption of the insulin receptor in its adipose tissue.12 Fat Insulin Receptor Knockout (“FIRKO”) mice have essentially normal appearance, food intake and fertility, are lean, insulin sensitive, and outlive normal (wild-type) animals by approximately 18%. Moreover, these animals are resistant to age-related and high fat diet-induced obesity and glucose intolerance.”
LONG TERM KETOGENIC DIET PROMOTES CANCER GROWTH
In spite of existing opinions about the anticancer actions of a ketogenic diet, we have shown that this anti-seizure therapy, especially in its long term usage, leads to excessive tumor growth. Prolonged feeding of a ketogenic diet promotes the growth of renal tumors by recruiting ERK1/2 and mTOR which are associated with the accumulation of oleic acid and the overproduction of growth hormone. Simultaneously, we observed that Nrf2, p53 and 8-oxoguanine glycosylase α dependent antitumor mechanisms were launched by the ketogenic diet. However, the pro-cancerous mechanisms finally took the ascendency by boosting tumor growth.
https://www.nature.com/articles/srep21807
rLIP76 inhibition (via FLAVON0IDS IN orange peel) prevents Obesity, Metabolic syndrome and cancer
“Feeding a Western high-fat diet (HFD) to C57BL/6 mice induces obesity, associated with a chronic inflammatory state, lipid transport, and metabolic derangements, and organ system effects that particularly prominent in the kidneys. Here, we report that RLIP76 homozygous knock-out (RLIP76−/−) mice are highly resistant to obesity as well as these other features of metabolic syndrome caused by HFD. The normal increase in pro-inflammatory and fibrotic markers associated with HFD induced obesity in wild-type C57B mice was broadly and nearly completely abrogated in RLIP76−/− mice. This is a particularly striking finding because chemical markers of oxidative stress including lipid hydroperoxides and alkenals were significantly higher in RLIP76−/− mice. Whereas HFD caused marked suppression of AMPK in wild-type C57B mice, RLIP76−/−. The baseline renal function was reduced in mice had baseline activation of AMP-activated protein kinase, which was not further affected by HFDRLIP76−/− mice as compared with wild-type, but was unaffected by HFD, in marked contrast to severe renal impairment and glomerulopathy in the wild-type mice given HFD. Our findings confirm a fundamental role of RLIP76 in regulating the function of obesity-promoting pro-inflammatory cytokines, and provide a novel mechanism for targeted therapy of obesity and metabolic syndrome.”https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3743508/
“When you get rid of this [RLIP76] gene in a mouse, it would appear that the mouse can’t get obese, it can’t get diabetes, it can’t get high cholesterol and it can’t get cancer,”explained Sanjay Awasthi, M.D., professor in the Division of Molecular Diabetes Research at City of Hope hospital.
Our recently published studies demonstrate that RLIP76−/− mice used for these studies were found to have marked insulin-sensitivity, and blood glucose was 46% lower than in RLIP76+/+animals (p<0.001). RLIP76−/− mice also had lower total serum cholesterol and triglycerides (43% and 40% of control, respectively; p<0.01) [1]. The hypoglycemia in RLIP76−/− mice is particularly remarkable because markers of oxidative-stress are remarkably increased in the tissues of the RLIP76−/− animals [1], [23]–[25]. Thus, in the absence of RLIP76, increases in these lipid-peroxidation products are insufficient by themselves to turn on any signaling pathway that can increase BG or lipids. Increased gluconeogenesis was particularly remarkable given that the activity of key gluconeogenic enzymes, G6Pase, F1,6-BPase, and PEPCK, in liver of RLIP76−/− mice was significantly inhibited.
Enhanced basal pAMPK levels in RLIP76−/− mice was another salient finding which strengthens the postulate that RLIP76 is a highly effective target for developing interventional strategies for MSy. Resveratrol, commonly used anti-oxidant, is known to activate AMPK which could contribute to its protective effects from high fat diet induced insulin-resistance [53], [54]. AMPK protects cells from stresses that cause ATP depletion by switching off ATP-consuming biosynthetic pathways. AMPK is activated by phosphorylation by an upstream protein kinase known as AMPK kinase. Activated AMPK can phosphorylate and regulate in vivo HMG-CoA, which is key regulatory enzyme of sterol synthesis [43], [47]. HMG-CoA limits the rate of cholesterol synthesis in liver tissue. Lipitor, inhibitor of HMG-CoA, exerts anti-inflammatory effects by lowering plasma cholesterol. Activation of AMPK leads to the inhibition of cholesterol synthesis by the phosphorylation of HMG-CoA reductase [43]. Loss of RLIP76 significantly affects the activation of stress and apoptosis pathway proteins [1], [25], [35]. Activation of AMPK leads to the inhibition of cholesterol synthesis by the phosphorylation of HMGCR. AMPK activation would be a good approach to treat T2D. These medications generally function to increase the effectiveness of insulin-mediated postprandial inhibition of hepatic gluconeogenesis. These findings provide a new insight on the mechanisms of action of hypoglycemic and/or hypolipidemic drugs.
RLIP76 knock-out mice survive well and are active. In our extensive and previously published studies, RLIP76 inhibition specifically leads to targeting signaling of importance in diabetes mellitus and other oxidative stress related conditions like cancers where targeting RLIP76 leads to selective cancer cell death without affecting the survival of normal cells and tissues [1],[31]–[33]. Hence, both global and selectively targeted approaches can be reasonably pursued as required while targeting RLIP76. In conclusion, our results suggest that RLIP76 is a key effector controlled by multiple proteins known to regulate the metabolic abnormalities of diabetes and metabolic syndrome, and that in its absence drugs that target these proteins will fail to function. The specific events that regulate the transport-effector/clathrin-endocytosis activity of RLIP76 (i.e. phosphorylation of RLIP76 by JNK, Akt, AMPK) will be explored in the future studies.
2′-Hydroxyflavanone (a flavonoid in orange peel): A novel strategy for targeting breast cancer
Intake of citrus fruits is known to reduce the risk for incidence of breast cancer. Hence, we tested the efficacy of citrus flavonoid 2′-hydroxyflavanone (2HF) in breast cancer. 2HF inhibited survival, clonogenic ability, cell cycle progression and induced apoptosis in breast cancer cells. 2HF also decreased VEGF levels and inhibited migratory capacity of breast cancer cells. Administration of 2HF led to regression of triple-negative MDA-MB-231 tumors in the mice xenograft model. 2HF decreased the levels of RLIP76 both studies and MDA-MB-231 xenograft model of breast cancer.
Western blot and histopathological analyses of resected tumors showed a decline in the levels of survival and proliferation markers Ki67, pAkt, survivin, and cell cycle proteins CDK4 and cyclin B1. 2HF treatment led to inhibition of angiogenesis as determined by decreased VEGF levels and angiogenesis marker CD31 . 2HF reversed the pro-/anti-apoptotic ratio of BAX/BCL-2 by decreasing anti-apoptotic protein BCL-2 and increasing pro-apoptotic proteins BAX and BIM . 2HF also decreased the mesenchymal markers vimentin and fibronectin along with causing a parallel increase in pro-differentiation protein E-cadherin. Collectively, the ability of 2HF to decrease RLIP76, VEGF and regulate critical proliferative, apoptotic and differentiation proteins together provides strong rationale to further develop 2HF based interventions for targeting breast cancer.
AMP-activated protein kinase (AMPK) is a serine/threonine kinase that is implicated in the control of energy metabolism and is considered to be a molecular target for the suppression of obesity and the treatment of metabolic syndrome. Here, we identified and characterized nootkatone, a constituent of grapefruit, as a naturally occurring AMPK activator. Nootkatone induced an increase in AMPKα1 and -α2 activity along with an increase in the AMP/ATP ratio and an increase the phosphorylation of AMPKα and the downstream target acetyl-CoA carboxylase (ACC), in C2C12 cells. Nootkatone-induced activation of AMPK was possibly mediated both by LKB1 and Ca2+/calmodulin-dependent protein kinase kinase. Nootkatone also upregulated PPARγ coactivator-1α in C2C12 cells and C57BL/6J mouse muscle. In addition, administration of nootkatone (200 mg/kg body wt) significantly enhanced AMPK activity, accompanied by LKB1, AMPK, and ACC phosphorylation in the liver and muscle of mice.
Whole body energy expenditure evaluated by indirect calorimetry was also increased by nootkatone administration. Long-term intake of diets containing 0.1% to 0.3% (wt/wt) nootkatone significantly reduced high-fat and high-sucrose diet-induced body weight gain, abdominal fat accumulation, and the development of hyperglycemia, hyperinsulinemia, and hyperleptinemia in C57BL/6J mice. Furthermore, endurance capacity, evaluated as swimming time to exhaustion in BALB/c mice, was 21% longer in mice fed 0.2% nootkatone than in control mice. These findings indicate that long-term intake of nootkatone is beneficial toward preventing obesity and improving physical performance and that these effects are due, at least in part, to enhanced energy metabolism through AMPK activation in skeletal muscle and liver.
⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐⭐
“WHAT’S THE BEST MEAL BETWEEN DRY OR FLUID RESTRICTED FASTS FOR FASTEST FAT LOSS?“
THE INTERSTELLAR ANTI-OBESITY LONGEVITY SPICE/PEEL ARUGULA PARSLEY SALAD!!!!
RECIPE: 2 cups arugula (highest nitric oxide plant) , 1 cup fresh parsley, 1 oz (tbs) olive oil, 1/4 cup lemon juice, 1 tsp INTERSTELLAR SPICE, 1 tsp INTERSTELLAR PEEL? (https://theinterstellarplan.com/product/spiceandpeelcombo/), salt and pepper to taste if needed.
“Obesity is a risk factor for developing type 2 diabetes and cardiovascular disease and has quickly become a world-wide pandemic with few tangible and safe treatment options. While it is generally accepted that the primary cause of obesity is energy imbalance, i.e., the calories consumed are greater than are utilized, understanding how caloric balance is regulated has proven a challenge. Many “distal” causes of obesity, such as the structural environment, occupation, and social influences, are exceedingly difficult to change or manipulate. Hence, molecular processes and pathways more proximal to the origins of obesity—those that directly regulate energy metabolism or caloric intake—appear to be more feasible targets for therapy. In particular, nitric oxide (NO) is emerging as a central regulator of energy metabolism and body composition. NO bioavailability is decreased in animal models of diet-induced obesity and in obese and insulin resistant patients, and increasing NO output has remarkable effects on obesity and insulin resistance. This review discusses the role of NO in regulating adiposity and insulin sensitivity and places its modes of action into context with the known causes and consequences of metabolic disease.”
Catalase neutralizes the toxic effects of hydrogen peroxide.
Beef liver is one of the highest food sources of catalase—non-coincidentally, liver was one of Bruce Lee’s favorite dishes!
You WANT a lean agile muscular physique like Bruce Lee.
NOT the freak of nature, totally unhealthy hulk anabolic steroid artificially induced body building fake muscle mass. Abnormal, artificial muscle and mass comes at the expense of longevity by hyperactivating mTOR. No bueno!!!! All time stupid idea. Do you know any hulk sized super centenarians? Me neither. They don’t exist.
BLACK TEA: HYDROGEN PEROXIDE SCAVENGER
Green tea gets all the love but BLACK TEA is the more potent hydrogen peroxide scavenger.
“Aqueous extracts of green and black teas have been shown to inhibit a variety of experimentally induced animal tumors, particularly ultraviolet (UV) B light-induced skin carcinogenesis. In the present study, we compared the effects of different extractable fractions of green and black teas on scavenging hydrogen peroxide (H2O2), and UV irradiation-induced formation of 8-hydroxy 2′-deoxyguanosine (8-OHdG) in vitro. Green and black teas have been extracted by serial chloroform, ethyl acetate and n-butanol, and divided into four subfractions designated as GT1-4 for green tea and BT1-4 for black tea, respectively.
The total extracts from green and black teas exhibited a potent scavenging capacity of exogenous H2O2 in a dose-dependent manner. It appeared that the total extracts from black tea scavenged H2O2 more potently than those from green tea. When tested individually, the potency of scavenging H2O2 by green tea subfractions was: GT2 > GT3 > GT1 > GT4, whereas the order of efficacy for black tea was: BT2 > BT3 > BT4 > BT1. In addition, we demonstrated that total fractions of green and black teas substantially inhibited the induction of 8-OHdG in calf thymus by all three portions of UV spectrum (UVA, B and C). Consistent with the capacity of scavenging H2O2, the subfractions from black tea showed a greater inhibition of UV-induced 8-OHdG than those from green tea.
At low concentrations, the order of potency of quenching of 8-OHdG by green tea subfractions was: GT2 > GT3 > GT4 > GT1 and the efficacy of all subfractions became similar at high concentrations. All subfractions of the black tea except BT1 strongly inhibited UV-induced 8-OHdG and the order of potency was: BT2 > BT3 > BT4 > BT1. Addition of (-)-epigallocatechin gallate (EGCG), an ingredient of green tea extract, to low concentration of green and black tea extracts substantially enhanced the scavenging of H2O2 and quenching of 8-OHdG, suggesting the important role of EGCG in the antioxidant activities of tea extracts. The potent scavenging of oxygen species and blocking of UV-induced oxidative DNA damage may, at least in part, explain the mechanism(s) by which green/black teas inhibit photocarcinogenesis.” https://www.ncbi.nlm.nih.gov/pubmed/10401606/
AMPK, MITOCHONDRIA DYSFUNCTION, AGING
“Recent studies have demonstrated a strong relationship between aging-associated reductions in mitochondrial function, dysregulated intracellular lipid metabolism, and insulin resistance. Given the important role of the AMP-activated protein kinase (AMPK) in the regulation of fat oxidation and mitochondrial biogenesis, we examined AMPK activity in young and old rats and found that acute stimulation of AMPK-α2 activity by 5′-aminoimidazole-4-carboxamide-1-β-D-ribofuranoside (AICAR) and exercise was blunted in skeletal muscle of old rats.
Furthermore, mitochondrial biogenesis in response to chronic activation of AMPK with β-guanidinopropionic acid (β-GPA) feeding was also diminished in old rats. These results suggest that aging-associated reductions in AMPK activity may be an important contributing factor in the reduced mitochondrial function and dysregulated intracellular lipid metabolism associated with aging.”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1885964/
AMPK ACTIVATION AND INTENSE INTERVAL EXERCISE
“Brief intense interval exercise activates AMPK and p38 MAPK signaling and increases the expression of PGC-1 in human skeletal muscle
In summary, the present study showed that four 30-s bouts of all out cycling increased phosphorylation of AMPK 1, AMPK 2, and p38 MAPK immediately following exercise and the mRNA expression of PGC-1 after 3 h of recovery. Specific signaling through AMPK and p38 MAPK to PGC-1 may therefore explain in part the metabolic remodeling in- duced by intense interval exercise training, including mitochondrial biogenesis and an increased capacity for glucose and fatty acid oxidation.”
http://m.jap.physiology.org/content/106/3/929.full.pdf
LEUCINE ACTIVATES mTOR and thus increases protein synthesis so it can be used to accelerate muscle growth AT BEGINNING of exercise window: http://m.ajpendo.physiology.org/content/294/2/E392.short
FASTING AND CARDIO-PROTECTION
Background— Intermittent fasting (IF), a dietary regimen in which food is available only every other day, increases the life span and reduces the incidence of age-associated diseases in rodents. We have reported neuroprotective effects of IF against ischemic injury of the brain. In this study, we examined the effects of IF on ischemic injury of the heart in rats.
Methods and Results— After 3 months of IF or regular every-day feeding (control) diets started in 2-month-old rats, myocardial infarction (MI) was induced by coronary artery ligation. Twenty-four hours after MI, its size in the IF group was 2-fold smaller, the number of apoptotic myocytes in the area at risk was 4-fold less, and the inflammatory response was significantly reduced compared with the control diet group. Serial echocardiography revealed that during 10 weeks after MI (with continuation of the IF regimen), the left ventricular (LV) remodeling and MI expansion that were observed in the control diet group were absent in the IF group. In a subgroup of animals with similar MI size at 1 week after MI, further observation revealed less remodeling, better LV function, and no MI expansion in the IF group compared with the control group.
Conclusions— IF protects the heart from ischemic injury and attenuates post-MI cardiac remodeling, likely via antiapoptotic and antiinflammatory mechanisms.http://circ.ahajournals.org/content/112/20/3115.short
MORE: https://scholar.google.com/scholar?hl=en&q=fasting+cardio+protection+&btnG=&as_sdt=1%2C33&as_sdtp
NITRIC OXIDE TO REPAIR DAMAGED MITOCHONDRIA AND REVERSE METABOLIC SYNDROME
Nitric oxide repairs the damaged mitochondria which excretes toxic hydrogen peroxide in your body which grays your hair and rapidly ages you.
Drink your arugula and beet juice daily and before and after dry fasts!
(Beets and arugula are the highest natural sources of nitrates that convert through bacteria in saliva to nitic oxide)
It has been recently found that mitochondrial biogenesis and function are enhanced by nitric oxide (NO), which is a key signaling molecule in vascular homeostasis.
HORMESIS
What’s that? To understand dry fasting (deliberate dehydration and starvation) is to understand hormesis.
“That which doesn’t kill you makes you stronger.” — Nietzsche
“All things are poison and nothing is without poison; only the dose makes a thing not a poison.” —Paracelsus
Even water and oxygen in excess can be deadly; as can their absence.
What we are doing is deliberately stressing the body— to make it more resilient.
“Hormesis is a biological phenomenon whereby a beneficial effect (improved health, stress tolerance, growth or longevity) results from exposure to low doses of an agent that is otherwise toxic or lethal when given at higher doses.”
Too much dry fasting can kill just as over eating or over drinking can kill.
We are in a sense flexing our cells “muscles”; making them extremely adaptable and tough.
“No water? No food? No problem. Wake me up when we got a real crisis on our hands.”
All about hormesis here: https://scholar.google.com/scholar
***INSULIN BLOCKS SIRT1*** (not good)
SIRT1 stands for sirtuin (silent mating type information regulation 2 homolog) 1 (S. cerevisiae), referring to the fact that its sirtuin homolog (biological equivalent across species) in yeast (S. cerevisiae) is Sir2. SIRT1 is an enzyme that deacetylates proteins that contribute to cellular regulation (reaction to stressors, longevity).
A major cause of aging is thought to result from the cumulative effects of cell loss over time. In yeast, caloric restriction (CR) delays aging by activating the Sir2 deacetylase. Here we show that expression of mammalian Sir2 (SIRT1) is induced in CR rats as well as in human cells that are treated with serum from these animals. Insulin and insulin-like growth factor 1 (IGF-1) attenuated this response. SIRT1 deacetylates the DNA repair factor Ku70, causing it to sequester the proapoptotic factor Bax away from mitochondria, thereby inhibiting stress-induced apoptotic cell death. Thus, CR could extend life-span by inducing SIRT1 expression and promoting the long-term survival of irreplaceable cells.
http://science.sciencemag.org/content/305/5682/390.short
FASTING AND STEM CELL REGENERATION
• fasting downregulates a IGF-1/PKA pathway in stem cells
• Prolong fasting protects hematopoietic cells from chemotoxicity
• Prolonged fasting cycles promote HSC self-renewal to reverse immunosuppression
• Inhibition of IGF-1 or PKA signaling mimics the effects of prolonged fasting
Immune system defects are at the center of aging and a range of diseases. Here, we show that prolonged fasting reduces circulating IGF-1 levels and PKA activity in various cell populations, leading to signal transduction changes in long-term hematopoietic stem cells (LT-HSCs) and niche cells that promote stress resistance, self-renewal, and lineage-balanced regeneration. Multiple cycles of fasting abated the immunosuppression and mortality caused by chemotherapy and reversed age-dependent myeloid-bias in mice, in agreement with preliminary data on the protection of lymphocytes from chemotoxicity in fasting patients. The proregenerative effects of fasting on stem cells were recapitulated by deficiencies in either IGF-1 or PKA and blunted by exogenous IGF-1. These findings link the reduced levels of IGF-1 caused by fasting to PKA signaling and establish their crucial role in regulating hematopoietic stem cell protection, self-renewal, and regeneration.
http://ac.els-cdn.com/S1…/1-s2.0-S1934590914001519-main.pdf
METABOLIC TRIGGERS OF AUTOPHAGY
In isolated cells, autophagy is generally induced by limitations in ATP availability or a lack of essential nutrients, including glucose and amino acids (i.e. FASTING or KETOGENIC diet).
COLD THERMOGENESIS & MTOR
To withstand cold you MUST activate mTOR to maintain body temperature.
MTOR (ACTIVATION) IS REQUIRED FOR BROWN ADIPOSE TISSUE RECRUITMENT AND METABOLIC ADAPTATION TO COLD
https://www.nature.com/articles/srep37223
MTOR (ACTIVATED) IS A KEY MODULATOR OF AGING AND AGE RELATED DISEASE
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3687363/
RAPAMYCIN BLOCKS INDUCTION OF THE THERMOGENIC PROGRAM IN WHITE ADIPOSE TISSUE
Here, we report that rapamycin blocks the ability of β-adrenergic signaling to induce beige adipocytes and expression of thermogenic genes in white adipose depots. Rapamycin enhanced transcriptional negative feedback on the β3-adrenergic receptor. However, thermogenic gene expression remained impaired even when the receptor was bypassed with a cell-permeable cAMP analog, revealing the existence of a second inhibitory mechanism. Accordingly, rapamycin-treated mice are cold intolerant, failing to maintain body temperature and weight when shifted to 4°C.
http://diabetes.diabetesjournals.org/content/65/4/927
RAMADAN IS DAILY INTERMITTENT DRY FASTING
Why not apply this wise practice every day and not just Ramadan?
“Fasting in the month of Ramadan is ordained on the Muslim believers. Ramadan fasting has not only been spiritually beneficial but it has physical, psychological, social and health benefits.
Ramadan is a month of self regulation, process of self purification, truthfulness and self trainings with the hope that this training will last beyond the end of Ramadan. One advantage of fasting is that the poor are given attention and benefits from charity and the faithful practice of the concept of neighbourhood and hospitality.
The physiological effect of fasting includes lowering of blood sugar, lowering of cholesterol and lowering of the systolic blood pressure. In fact, Ramadan fasting would be an ideal recommendation for treatment of mild to moderate, stable, non-insulin diabetes, obesity and essential hypertension. Fasting is powerful therapeutic processes that can help people recover from mild to severe health conditions. Our body has a self healing power, in order to activate this power, the stomach must be kept empty, if Ramadan fasting is done properly it can help to recover from most diseases.”
http://www.omicsonline.org/psycho-social-behaviour-and-heal
P53: THE ANTI-TUMOR GENE AND FASTING
What is P53?
P53, also known as TP53 or tumor protein (EC :2.7.1.37) is a gene that codes for a protein that regulates the cell cycle and hence functions as a tumor suppression. It is very important for cells in multicellular organisms to suppress cancer.
“Our findings that CR or a 1 day/week fast suppressed carcinogenesis—even when started late in life in mice predestined to develop tumors due to decreased p53 gene dosage—support efforts to identify suitable interventions influencing energy balance in humans as a tool for cancer prevention.”
http://m.carcin.oxfordjournals.org/content/23/5/817.full
Ghrelin the anti-aging hunger hormone is released about 12 hours after your last meal. This is why you want lots of Ghrelin:
http://www.nature.com/…/jo…/vaop/ncurrent/pdf/mp2015220a.pdf
But what about lack of water and kidneys?
“High fluid intake does not appear to slow renal disease progression in humans.”
http://www.sciencedirect.com/…/article/pii/S0272638603001938
BAKING SODA INCREASES LIFESPAN
Klotho, a protein counteracting aging, is a powerful inhibitor of 1,25-dihydroxyvitamin D3 [1,25(OH)2D3] formation and regulator of mineral metabolism. In klotho hypomorphic (kl/kl) mice, excessive 1,25(OH)2D3 formation leads to hypercalcemia, hyperphosphatemia and vascular calcification, severe growth deficits, accelerated aging and early death. Kl/kl mice further suffer from extracellular volume depletion and hypotension, leading to the stimulation of antidiuretic hormone and aldosterone release. A vitamin D-deficient diet, restriction of dietary phosphate, inhibition of mineralocorticoid receptors with spironolactone, and dietary NaCl all extend the lifespan of kl/kl mice. Kl/kl mice suffer from acidosis. The present study explored whether replacement of tap drinking water by 150 mM NaHCO3 affects the growth, tissue calcification, and lifespan of kl/kl mice.
As a result, NaHCO3 administration to kl/kl mice did not reverse the growth deficit but substantially decreased tissue calcification and significantly increased the average lifespan from 78 to 127 days. NaHCO3 did not significantly affect plasma concentrations of 1,25(OH)2D3 and Ca2+ but significantly decreased plasma phosphate concentration and plasma aldosterone concentration. The present study reveals a novel effect of bicarbonate, i.e., a favorable influence on vascular calcification and early death of klotho-deficient mice.
http://www.physiology.org/doi/abs/10.1152/ajprenal.00037.2015
A CORTISOL FUNCTION IS TO RAISE PH
“The effect of cortisol on pH is even clearer if one considers the change in pH after injection, since in the afternoon urinary pH usually fell steadily …… After cortisol pH always rose, and only in one experiment with aldosterone was a comparable rise seen.”
http://onlinelibrary.wiley.com/doi/10.1113/jphysiol.1960.sp006440/pdf
With this knowledge you can logically conclude that if you are ACIDIC— you are releasing more cortisol.
BAKING SODA RAISES PH WITHOUT THE NEED FOR RELEASING CORTISOL.
“Mean daily plasma cortisol decreased from 264 ± 45 to 232 ± 43 nmol/l (P = 0.032), and urinary excretion of tetrahydrocortisol (THF) decreased from 2,410 ± 210 to 2,098 ± 190 μg/24 h (P = 0.027). No significant effect was found on free IGF-1, PTH/1,25(OH)2 vitamin D, or thyroid hormones. An acidogenic Western diet results in mild metabolic acidosis in association with a state of cortisol excess, altered divalent ion metabolism, and increased bone resorptive indices. Acidosis-induced increases in cortisol secretion and plasma concentration may play a role in mild acidosis-induced alterations in bone metabolism and possibly in osteoporosis associated with an acidogenic Western diet.“
http://www.physiology.org/doi/abs/10.1152/ajprenal.00212.2002
MATCHA BLOCKS CORTISOL
(dry fasting creates more cortisol via acidosis)
“In conclusion, we provide evidence that aqueous extracts of tea (Camellia sinensis) are able to inhibit cortisol formation by the enzyme 11β-HSD1. From several abundant constitutents of tea, the major phenolic compound EGCG could be identified as the most potent inhibitor of human 11β-HSD1, with inhibition constants of Ki = 22.68 µM in microsomes and Ki = 18.74 µM for the purified enzyme, respectively. The mechanism of EGCG inhibition is most likely a direct binding to the active site of 11β-HSD1, which is supported by enzyme kinetic studies and a computer aided docking model. Our results decipher the mechanism by which catechins such as EGCG, or green tea in general, have been successfully consumed for thousand of years for general health benefits. These polyphenolic compounds may serve as model structures for the development of novel agents to treat the metabolic syndrome and related diseases.”
PROTECT KIDNEYS by ALKALINIZING
Periodically before and after dry fasts with a simple teaspoon of BAKING SODA in water.
http://onlinelibrary.wiley.com/doi/10.1002/1873-3468.12359/full
WHY YOU WANT TO TAKE BAKING SODA TO RAISE PH WHILE IN KETOSIS FOR MAXIMUM FAT LOSS
“An acidosis produced by feeding HCl is antiketogenic and an alkalosis resulting from NaHC03 (baking soda) administration is ketogenic in the fasting albino rat when ketosis is measured by the level of the ketone bodies in the blood as well as the degree of ketonuria. Acid administration causes an increase in the liver glycogen concentration and in the daily excretion of nitrogen and sulfur in the urine, while the alkalosis leads to a decrease from normal in all of these values. It is concluded from this that the antiketogenic effect of acidosis depends upon an increase in protein catabolism and an increase in glucose formation therefrom, while alkalosis is ketogenic because it has an opposite effect upon this process.”
We want MORE ketosis (fat burning) not less!
IMPORTANT: Once your ph drops you are eating muscle
*First attain deep level ketosis from dry fasting then add baking soda; strive to keep urine around 7.5ph while simultaneously maintaining deep level ketosis (80-160).
“Oral Hydration and Alkalinization is Noninferior to Intravenous Therapy for Prevention of Contrast-Induced Nephropathy in Patients with Chronic Kidney Disease”
http://onlinelibrary.wiley.com/…/j.1540-8183.2010.0058…/full
More on WHY YOU WANT TO BE ALKALINE while in KETOSIS.
(KETO-ALKALINE) Acidic ph inhibits autophagy; alkaline stimulates it.
“In this study, we show that in rat cardiomyocytes, acidic extracellular pH (pHe) inhibits autophagy, whereas alkaline pHe stimulates it. We also find that adenosine monophosphate-activated protein kinase (AMPK), mammalian target of rapamycin (mTOR) and Unc-51-like kinase 1 (ULK1) are very sensitive to pHe changes. Furthermore, by interfering with AMPK, mTOR or ULK1 activity, we demonstrate that the AMPK–ULK1 pathway, but not the mTOR pathway, plays a crucial role on pHe-regulated autophagy and cardiomyocyte viability. These data provide a potential therapeutic strategy against cardiomyocyte injury triggered by pH fluctuations.” http://onlinelibrary.wiley.com/doi/10.1002/1873-3468.12359/full
HEADACHES are common on the first day for new dry fasters and those not experienced with KETOSIS. It usually takes place around the 18 hour mark and goes away after a few hours. This is the brain switching off glucose and now using ketones for fuel. The headache could also be symptomatic of caffeine withdrawal. Totally normal; push through, it passes.
50% INCREASE IN LIFESPAN BY COMPLETE ABSENCE OF FOOD (study)
“Remarkably, the greatest increase in lifespan was observed …maintained in the complete absence of food, with a 50% increase in median and maximum lifespan relative to control-fed animals.”
http://onlinelibrary.wiley.com/…/j.1474-9726.2006.0023…/full
INCREASES LIFESPAN UP TO 60% (study)
“Dietary Restriction (DR) increases the rodent lifespan by up to 60% in part by delaying the occurrence of many chronic diseases, and in part by slowing the rate of biological or physiological aging (1).
Among its many protective effects of DR described in mice, the most notable include those on increasing insulin sensitivity and on the attenuation of B amyloid deposition in a model for Alzheimer’s Disease (54). Severe dietary restriction or STARVATION (dry fasting) may also be applicable to disease treatment. For example, fasting protects mice against high dose chemotherapy in part by reducing serum IGF-I signalling but does not protect cancer cells, since oncogene mutations prevent the activation of the stress resistance pathways in response to the reduction in glucose, IGF-I and other growth factors caused by nutrient deprivation (55).”
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3607354/
“The higher the metabolism speed, the shorter the lifespan, and vice versa.”
YOU DO NOT WANT A FAST METABOLISM
http://en.cnki.com.cn/Article_en/CJFDTOTAL-SPKX201315076.htm
Over 24 weeks, a low-carbohydrate diet program led to greater weight loss, reduction in serum triglyceride level, and increase in HDL cholesterol level compared with a low-fat diet.
THE STARVATION TREATMENT OF DIABETES
ADHD
Adiponectin is associated with longevity and heightened insulin sensitivity and has anti cancer properties; it is a hormone excreted primarily from adipose tissue(fat). Fasting is exceptional for releasing Adiponectin and preventing adhd not to mention inhibiting disease and prolonging life.
http://www.psy-journal.com/a…/S0165-1781(14)00061-4/abstract
Centenarians and high Adiponectin levels
FGF21 (the starvation hormone)
Fibroblast growth factor-21 (FGF21) is a hormone secreted by the liver during fasting that elicits diverse aspects of the adaptive starvation response. Among its effects, FGF21 induces hepatic fatty acid oxidation and ketogenesis, increases insulin sensitivity, blocks somatic growth and causes bone loss. Here we show that transgenic overexpression of FGF21 markedly extends lifespan in mice without reducing food intake or affecting markers of NAD+ metabolism or AMP kinase and mTOR signaling. Transcriptomic analysis suggests that FGF21 acts primarily by blunting the growth hormone/insulin-like growth factor-1 signaling pathway in liver. These findings raise the possibility that FGF21 can be used to extend lifespan in other species.
FGF21 is specifically induced by HMGCS2 activity. The oxidized form of ketone bodies (acetoacetate) in a cultured medium also induced FGF21, possibly via a SIRT1-dependent mechanism.[3] HMGCS2 activity has also been shown to be increased by deacetylation of lysines 310, 447, and 473 via SIRT3 in the mitochondria.[4]
While FGF21 is expressed in numerous tissues, including liver, brown adipose tissue, white adipose tissue and pancreas, circulating levels of FGF21 are derived specifically from the liver in mice.[5] In liver FGF21 expression is regulated by PPARα and levels rise substantially with both FASTING and consumption of ketogenic diets.
http://elifesciences.org/content/1/e00065v1
FULVIC ACID and the importance of minerals
Watch the light bulb demonstration: Www.purefulvicminerals.com
Your body is just like a battery; no minerals—no electricity. “All life is Electric”
A CONCEPTUAL REVOLUTION OF BIBLICAL PROPORTIONS
“The previously unknown capacity of human body to uses water as source of electrons is an astonishing finding that is a conceptual revolution of biblical proportions. The sacred role of glucose as source of energy now is broken into small pieces. Therefore human body build up its biomass arising from glucose, thereby carbohydrates (or meals) are just a source of carbon chains of different lengths, branching; combinations with other elements and so forth. We could say that glucose (C6 H12O6 ) is the perfect building block. Our body is able to synthesize even nucleic acids with it. But our body fulfills its energy needs by means of the unexpected and astonishing capacity of melanin to split and re-form the water molecule. “
http://www.omicsonline.org/human-photosynthesis-a-turning-p
For constipation or bowel issues:
1TBS of Milk of Magnesia should do the trick
AMPK ACTIVATORS & MTOR INHIBITORS
At the start or end of your dry fast, or as a brief interval for instance in mornings, to increase mTOR inhibition, AMPK activation and maintain autophagy effects do a 4-6 oz cup of BLACK (no cream or mct oil as this releases insulin) coffee, matcha, black tea, INTERSTELLAR BLEND. This addition will provide energy and clarity all day long without engaging mTOR pathway, halting autophagy or deactivating AMPK — plus you won’t be hungry or craving anything. If you research the anti aging herbs in the blend you will see why this is a wise decision to add to your daily regiment and if you choose to drink it upon rising or as an intermission you are maintaining calorie (carb/protein) restriction (mTOR inhibition/AMPK activation/autophagy) until feeding window. You can also skip this altogether and drink this at feeding window and dry fast (or fluid restrict ie. a second small 4 0z cup mid afternoon is not a deal breaker) straight through the 18-22 hours. ZERO CALORIES. Explore both options to find best fit. You want a plan that you can stick to and that provides results.
A few of the herbs in INTERSTELLAR blends that inhibit mTOR:
Reishi
“Thus, herein we define the mechanistic effects of Reishi focusing on the phosphoinositide-3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) pathway, a regulator of cell survival and growth. The present study demonstrates that Reishi treated IBC SUM-149 cells have reduced expression of mTOR downstream effectors at early treatment times, as we observe reduced eIF4G levels coupled with increased levels of eIF4E bound to 4E-BP, with consequential protein synthesis reduction.”
http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0057431
Astragalus
“Further adding to our recent suggestion on its correlation with phosphatidylinositol 3-kinase (PI3K)-Akt signaling, we have now revealed that AST caused overexpression of PTEN and down-regulation of mammalian target of rapamycin (mTOR) expression. Nevertheless, these events were preceded by a decrease in nuclear factor-κB (NF-κB)/DNA binding activity with continuous ERK 1/2 activation. Some of these effects became more intense in cytokine-induced cells. Our findings in this study suggest that AST induces the extrinsic apoptotic cascade and causes cell cycle arrest in HT-29 cells by modulation of both mTOR and ERK signaling pathways, of which inhibition of NF-κB is important in the latter mechanism. Most of the above processes are more pronounced in cytokine-induced cells.”
https://www.spandidos-publications.com/ijmm/26/3/341?text=abstract
He Shou Wu (Polygonum multiflorum)
“Twelve-week-old male obese Zucker diabetic fatty (ZDF) rats were used. Compared with their lean counterparts, obese ZDF rats exhibited hypertension and mesenteric artery endothelial dysfunction (n=6, P<0.05), along with impaired Akt/mechanistic target of rapamycin (mTOR) signaling and upregulated expression of beclin1, LC3II/I, p62, ATG5 and ATG7 in mesenteric arterioles (n=6, P<0.05), suggesting increased and impeded autophagy in mesenteric arteries from ZDF rats. Two-week TSG administration (100mg/kg/day) by gavage significantly decreased blood pressure (BP) and improved microvascular endothelial function (n=6, P<0.05), reactivated Akt/mTOR pathway and decreased endothelial autophagy in obese ZDF rats (n=6, P<0.05). Rapamycin pretreatment blocked the hypotensive effect of TSG in obese ZDF rats. Suppression of Akt and mTOR expression with siRNAs significantly blunted the anti-autophagic effect of TSG in cultured human umbilical vein endothelial cells as evidenced by abnormal autophagic flux and increased expression of autophagy-associated proteins, respectively (n=3, P<0.05).”
Rhodiola
“In addition to its multiplex stress-protective activity, Rhodiola rosea extracts have recently demonstrated its anti-aging, anti-inflammation, immunostimulating, DNA repair, and anti-cancer effects in different model systems. Molecular mechanisms of Rhodiola rosea extracts’ action have been studied mainly along with one of its bioactive compounds, salidroside. BothRhodiola rosea extracts and salidroside have contrast molecular mechanisms on cancer and normal physiological functions. For cancer, Rhodiola rosea extracts and salidroside inhibit the mTOR pathway and reduce angiogenesis through downregulation of the expression of HIF-1α/HIF-2α.”
https://link.springer.com/article/10.1007/s40495-017-0106-1
Ginseng
“Derived from Panax ginseng, the natural product 20(S)-Protopanaxadiol (PPD) has been reported for its cytotoxicity against several cancer cell lines. The molecular mechanism is, however, not well understood. Here we show that PPD significantly inhibits proliferation, induces apoptosis and causes G2/M cell cycle arrest in human laryngeal carcinoma cells (Hep-2 cells). PPD also decreases the levels of proteins related to cell proliferation. Moreover, PPD-induced apoptosis is characterized by a dose-dependent down-regulation of Bcl-2 expression and up-regulation of Bax, and is accompanied by the activation of Caspase-3 as well. Further molecular mechanism is revealed by direct stochastic optical reconstruction microscopy (dSTORM)—a novel high-precision localization microscopy which enables effective resolution down to the order of 10 nm. It shows the expression and spatial arrangement of mTOR and its downstream effectors, demonstrating that this ginsenoside exerts its excellent anticancer effects via down-regulation of mTOR signaling pathway in Hep-2 cells. Taken together, our findings elucidate that the antitumor effect of PPD is associated with its regulation of mTOR expression and distribution, which encourages further studies of PPD as a promising therapeutic agent against laryngeal carcinoma.”
http://www.mdpi.com/1420-3049/22/3/486/htm
A few of the herbs in INTERSTELLAR blends that activate AMPK:
Gynostemma
“Gynostemma pentaphyllum (G. pentaphyllum) is a herbaceous vine of the family Cucurbitaceae (cucumber or gourd family) that is indigenous to and widely used in Asian countries including Korea, China, and Japan as traditional medicines or tea. Total extracts or saponins from this plant have been shown to exert a wide range of beneficial effects such as reducing cholesterol and blood glucose levels, strengthening immunity, and inhibiting cancer growth ([13-16]). These activities likely overlap with diverse downstream effects on AMPK activation ([9]). In vitro studies revealed that damulin A and B, two dammarane-type saponins purified from the leaves of G. pentaphyllum, are able to increase the phosphorylation of AMPK and acetyl-CoA carboxylase (ACC) through which β-oxidation can be stimulated ([17]).
We recently reported that, the ability of a G. pentaphyllum leaf ethanol extract to activate AMPK is increased by autoclaving with increasing the levels of AMPK activators damulin A and B ([18]). In both cultured HepG2 and L6 myotube cells, this heat-processed G. pentaphyllum extract named “actiponin” dose-dependently increases the expression of key factors that regulate fat oxidation and adaptive thermogenesis while decreasing the expression of lipogenic transcription factors ([18]). Furthermore, oral administration of actiponin reduces body fat mass in ob/ob mice by stimulating AMPK and ACC phosphorylation in the soleus muscle ([18]). Although the antiobesity activities of G. pentaphyllum extract have been demonstrated in vitro and in vivo ([17, 18]), the effects of this plant extract on humans are unclear. Therefore, we performed the first clinical trial for evaluating the antiobesity effects of actiponin on human subjects. The objective of the present study was to document the effect of 12-week actiponin supplementation on body fat composition (particularly abdominal fat) in obese Korean participants using a randomized, double-blind, placebo-controlled protocol.”
http://onlinelibrary.wiley.com/doi/10.1002/oby.20539/full
Ginseng
“Korean Red Ginseng attenuates ethanol-induced steatosis and oxidative stress via AMPK/Sirt1 activation
These results demonstrate that RGE and its ginsenoside components inhibit alcoholic steatosis and liver injury by adenosine monophosphate-activated protein kinase/sirtuin 1 activation both in vivo and in vitro, suggesting that RGE may have a potential to treat alcoholic liver disease.”
“Anti-diabetic effect of black ginseng extract by augmentation of AMPK protein activity and upregulation of GLUT2 and GLUT4 expression in db/db mice
The mice group treated with GBG05-FF showed decreased fasting blood glucose and glucose tolerance compared to that of the nontreated GBG05-FF group. In the blood analysis, GBG05-FF decreased main plasma parameter such as HbA1c, triglyceride, and total-cholesterol levels related to diabetes and improved the expression of genes and protein related to glucose homeostasis and glucose uptake in the liver and muscle. The histological analysis result shows that GBG05-FF decreased lipid accumulation in the liver and damage in the muscle. Moreover, GBG05-FF increased the phosphorylation of the AMPK in the liver and upregulated the expression of GLUT2 in liver and GLUT4 in muscle. Therefore, the mechanisms of GBG05-FF may be related to suppressing gluconeogenesis by activating AMPK in the liver and affecting glucose uptake in surrounding tissues via the upregulation of GLUT2 and GLUT4 expression.” https://bmccomplementalternmed.biomedcentral.com/articles/10.1186/s12906-017-1839-4
“Panax ginseng extract regulates autophagy by AMPK signaling in A549 human lung cancer cells
Panax ginseng has been used worldwide as a traditional medicine for the treatment of cancer and other diseases. The antiproliferative activity of ginseng has been increased after enzymatic processing of ginseng saponin, which may result in the accumulation of minor saponins, such as Rh2, Rg3, compound K and protopanaxatriol type (PPT) in modified regular ginseng extract (MRGX). In the present study, the anticancer activity and the associated mechanisms of MRGX were investigated using A549 human lung cancer cells. To elucidate the mechanisms underlying the effects of MRGX, we performed a microarray analysis of gene expression in the A549 cells. Molecular mechanisms that were associated with the anticancer activity of MRGX were studied, with a special focus on the autophagy-related multiple signaling pathways in lung cancer cells. Microarray analyses elucidated autophagy-related genes affected by MRGX. Administration of MRGX at 100 µg/ml induced punctate cytoplasmic expression of LC3, Beclin-1 and ATG5 and increased expression of endogenous LC3-II whereas 50 µg/ml did not inhibit the proliferation of A549 cells. Compared to the control cells, in cells treated with MRGX at 100 µg/ml, the level of p-Akt was increased, while that of mTOR-4EBP1 was decreased. Downregulation of mTOR and 4EBP1 in the MRGX-treated cells was found not to be a p-Ulk (S757)-dependent pathway, but a p-Ulk (S317)-dependent autophagic pathway, using AMPK. These data suggest that MRGX regulates AMPK and induces autophagy in lung cancer cells.”
https://www.spandidos-publications.com/or/37/6/3287?text=fulltext
“Effects of High Hydrostatic Pressure Extract of Korean Fresh Ginseng on Hepatic Lipid Accumulation and AMPK Activation in HepG2 Cells
Ginseng is widely used as a medicinal herb and has demonstrated effects against liver diseases. The aim of this study is to investigate the hypolipidemic effects of the high hydrostatic pressure extract of Korean fresh ginseng (HEG) on hepatic lipid accumulation in HepG2 cells. The intracellular triglyceride and cholesterol contents were determined using enzymatic colorimetric methods. The mRNA levels of fatty acid synthase (FAS) and 3- hydroxy-3-methyl-glutaryl CoA reductase (HMGCR) were assayed by quantitative real-time PCR. The activity of AMP-activated protein kinase (AMPK) was measured with an AMPK kinase assay kit. HEG significantly reduced hepatic triglyceride and cholesterol contents in HepG2 cells. Furthermore, HEG suppressed the expression of FAS, a key enzyme in fatty acid synthesis, and HMGCR, a rate-limiting enzyme in hepatic cholesterol synthesis. Additionally, HEG increased the activity of AMPK, a major regulator of lipid metabolism. These results suggest that HEG reduces hepatic lipid accumulation with inhibition of FAS and HMGCR expression and stimulation of AMPK activity in HepG2 cells. Consequently, HEG may be beneficial as a functional food ingredient to improve various hepatic diseases by reducing hepatic lipid accumulation.”
https://pdfs.semanticscholar.org/8c06/411ec6ee5bbe1ddf159cde3c9d185171af05.pdf
Reishi Mushroom
“ReishiMax, mushroom based dietary supplement, inhibits adipocyte differentiation, stimulates glucose uptake and activates AMPK
RM inhibited adipocyte differentiation through the suppresion of expression of adipogenic transcription factors peroxisome proliferator-activated receptor-γ (PPAR-γ), sterol regulatory element binding element protein-1c (SREBP-1c) and CCAAT/enhancer binding protein-α (C/EBP-α). RM also suppressed expression of enzymes and proteins responsible for lipid synthesis, transport and storage: fatty acid synthase (FAS), acyl-CoA synthetase-1 (ACS1), fatty acid binding protein-4 (FABP4), fatty acid transport protein-1 (FATP1) and perilipin. RM induced AMP-activated protein kinase (AMPK) and increased glucose uptake by adipocytes.”
https://bmccomplementalternmed.biomedcentral.com/articles/10.1186/1472-6882-11-74
Ganoderma Lucidum
“Ganoderma lucidum polysaccharides improves cerebral infarction by regulating AMPK/eNOS signaling
Recent studies have shown that ganoderma lucidum polysaccharides (GLP) are characterized by immune regulation and anti-tumor functions. However, little research has been conducted to evaluate their effects on cerebral ischemia. In the current study, we first showed that GLP treatment improved neurological deficits, cerebral infarct volume, and brain edema rate, indicating the protective role of GLP in middle cerebral artery occlusion (MCAO) in rats. Furthermore, GLP treatment can significantly increased the cerebral vascular density in the ischemic area as well as angiogenesis after cerebral ischemia. Moreover, after cerebral ischemia, Nestin positive cells in the subventricular zone (SVZ) area were significantly decreased, while the number of Nestin positive cells was significantly increased after administration of GLP. Western blot analysis showed that increased phosphorylation of Amp-activated protein (AMP) kinase (AMPK) and eNOS after cerebral ischemia, and GLP treatment can further enhanced the phosphorylation of AMPK and eNOS in rat brains. In summary, GLP improved cerebral infarction mainly by enhancing the activation of AMPK and eNOS, thereby prompting angiogenesis and neuron regeneration.”
http://ijcem.com/files/ijcem0056954.pdf
Lycium Barbarum (Goi)
“Lycium barbarum Suppress SREBP-1c Expression and Prevent Diet-Induced Fatty Liver through AMPK Activation
Lycium barbarum polysaccharide (LBP) is well known in traditional Chinese herbal medicine that, has beneficial effects. Previous study reported that LBP reduced blood glucose and serum lipids. However, the underlying LBP-regulating mechanisms remain largely unknown. The main purpose of this study was to investigate whether LBP prevented fatty liver through activation of adenosine monophosphate-activated protein kinase (AMPK) and suppression of sterol regulatory element-binding protein-1c (SREBP-1c). Male C57BL/6J mice were fed a low-fat diet, high-fat diet, or 100 mg/kg LBP-treatment diet for 24 weeks. HepG2 cells were treated with LBP in the presence of palmitic acid. In our study, LBP can improve body compositions and lipid metabolic profiles in high-fat diet-fed mice. Oil Red O staining in vivo and in vitro showed that LBP significantly reduced hepatic intracellular triacylglycerol accumulation. H&E staining also showed that LBP can attenuate liver steatosis. Hepatic genes expression profiles demonstrated that LBP can activate the phosphorylation of AMPK, suppress nuclear expression of SREBP-1c, and decrease protein and mRNA expression of lipogenic genes in vivo or in vitro. Moreover, LBP significantly elevated uncoupling protein-1 (UCP1) and peroxisome proliferator-activated receptor- coactivator-1 (PGC-1) expression of brown adipose tissue. In summary, LBP possesses a potential novel treatment in preventing diet-induced fatty liver.”
https://www.hindawi.com/journals/bmri/2014/196198/abs/
Bitter Melon
REMEMBER: The less you drink the faster the fast works so if you do the morning drink keep it to 4-6 oz. Don’t ingest anything else until feeding/exercise window.
**************************************
FOR LONGER DRY FASTS 3+ DAYS (preparation)
**************************************
1. ALKALINIZE
As the dry fast progresses urine ph will drop and continue to drop until you break fast. (Ketone bodies are acidic in nature.)
ALWAYS, ALWAYS, ALWAYS alkalinize urine before and again after TO PROTECT KIDNEYS AND AVOID KIDNEY STONES. This is easily accomplished by drinking 1 teaspoon of baking soda in 8oz water.
*IF you foolishly skip this step don’t cry about getting a kidney stone or having kidney pain in the group as you were STRONGLY WARNED TO ALKALINIZE and dissolve all acid formed crystals in urinary tract before hand.
WHY USE BAKING SODA FOR PREVENTING KIDNEY STONES OR KIDNEY PROBLEMS?
Because it works and fast!
I recommend raising urine ph to an 8 NOT 7 as in study before long dry fasts and once again after.
PH STRIPS TO TEST URINE: http://www.amazon.com/…/…/B00LY1KIWY/ref=zg_bs_3013605011_10
Conclusions: Bicarbonate therapy remains an attractive option for the treatment of radiolucent kidney stones. The presence of hyperuricaemia or hyperuricosuria appears to influence the success rate. Further prospective randomised studies are needed to identify the most tolerable and effective treatment regime as well as the optimal duration of treatment. Dual-energy CT may hold the key to identifying patients most likely to benefit from treatment.
http://m.uro.sagepub.com/…/…/02/10/2051415816631856.abstract
2. CELLULAR ELECTRICITY
I recommend “charging” cells with trace minerals via Fulvic acid. Use either Shilajit or Fulvic mineral source. Without electricity you have no energy. WATCH light bulb demo: http://www.purefulvicminerals.com/product-comparison
3. AMPK ACTIVATORS/ MTOR INHIBITORS
I always utilize before hand to jump start the process SUPERTONIC HERBS that activate AMPK and inhibit mTOR and promote autophagy like what’s in Interstellar Blend. www.interstellarblend.com
4. THIRST AND HEAT
The body heats up the longer you go because the cells are incinerating junk via autophagy. Thirst may become intense. I recommend ICE BATHS & ICE SHOWERS to cool down and keep going. You will find this blissfully rejuvenating.
5. STUDY THIS POST AND THE LINKS. The more informed you are the less fear you will have and the greater resolve to not quit.
6. DEHYDRATION WORRIES
Skin tenting purportedly begins at 15% body water loss. At 7 days I had zero skin tenting still. NOBODY in group is in danger of dehydration in 72 hours. Nobody. Unless maybe you are in Death Valley, CA in 120+ degree heat sitting there sweating.
****IF YOU GOT FAT YOU GOT WATER.****
So have ZERO FEAR of going 3 days your first time. Alkalinize and launch!
What is skin tenting or skin turgor test???
https://www.google.com/search…
BLOOD PRESSURE CONCERNS
Be not overly concerned by blood pressure irregularities; the body as it acclimates itself to survival conditions corrects itself the longer you go. It may fluctuate up and down the first 3 days but by day 4 and onward it stabilizes.
Blood pressure and resting pulse without fail on all extended dry fasts (beyond 3 days) goes down and this is very positive.
Have no fear or worries; perfectly natural process.
HUMAN PHOTOSYNTHESIS
So the real source of energy of the eukaryotic cell is water, so the sacred role of glucose as an energy source now breaks into a thousand pieces. We ended up saying that if the energy source was glucose, diabetic patients would fly.
The human body with four billion years of evolution is far beyond our ability to abstraction, but in origin the body is relatively simple: everything comes, everything is soaked, and everything is governed by photosynthesis, both in plants and in us.
http://www.ncbi.nlm.nih.gov/…/P…/pdf/1878-5085-5-S1-A146.pdf
MELANIN THE HUMAN CHLOROPHYLL
“The main source of energy of the eukaryotic cell is water, not ATP. The profound misconception that food, glucose or ATP are the main source of energy has its basis in the lack of knowledge of the hitherto unknown capacity of melanin to split the water molecule [5]. Until today, the fact that human tissues have the capability to take hydrogen from water—the energy carrier by excellence in the whole Universe—arising from the splitting of water, as plants do, was totally unknown before our work.It was unthinkable that an expensive chemical reaction, from the energetic point of view, such as water dissociation, that requires 2000˚C in the laboratory to take place, might occur at room temperature in our body. The sole possibility seemed berserk. However, our studies researching the three main causes of blindness allowed us to detect the hitherto unknown fact that melanin is the “human chlorophyll”. This amazing compound absorbs photonic energy and transforms it into chemical energy.”
http://file.scirp.org/Html/7-2400067_7404.htm#%23%23
GLUCONEOGENESIS FROM FAT
“That sugar can be converted into fatty acids in humans is a well-known fact. The question whether the reverse direction, i.e., gluconeogenesis from fatty acids, is also feasible has been a topic of intense debate since the end of the 19th century. With the discovery of the glyoxylate shunt that allows this conversion in some bacteria, plants, fungi and nematodes it has been considered infeasible in humans since the corresponding enzymes could not be detected. However, by this finding only a single route for gluconeogenesis from fatty acids has been ruled out. To address the question whether there might exist alternative routes in humans we searched for gluconeogenic routes from fatty acids in a metabolic network comprising all reactions known to take place in humans.
****Thus, we were able to identify several pathways showing that this conversion is indeed feasible.****
Analyzing evidence concerning the detected pathways lends support to their importance during times of starvation, fasting, carbohydrate reduced and ketogenic diets and other situations in which the nutrition is low on carbohydrates. Moreover, the energetic investment required for this pathway can help to explain the particular efficiency of carbohydrate reduced and ketogenic diets such as the Atkins diet.”
http://journals.plos.org/ploscompbiol/article/asset
HEAT ON DRY FASTS
Sauna, hot tub, sun exposure, steam room, intense exercise causing profuse sweating NOT ADVISED on long dry fasts— over 24 hours. Under 24 hours it can be used to speed up ketosis induction.
KETONES IDEAL FUEL FOR BODY
“Ketones are the ideal fuel for our bodies unlike glucose – which is damaging, less stable, more excitatory and in fact shortens your life span. Ketones are non-glycating, which is to say, they don’t have a caramelizing ageing effect on your body. A healthy ketosis also helps starve cancer cells as they are unable to use ketones for fuel, relying on glucose alone for their growth.
The energy producing factories of our cells – the mitochondria – work much better on a ketogenic diet as they are able to increase energy levels in a stable, long-burning, efficient, and steady way. Not only that, a ketogenic diet induces epigenetic changes which increases the energetic output of our mitochondria, reduces the production of damaging free radicals, and favours the production of GABA – a major inhibitory brain chemical. GABA has an essential relaxing influence and its favored production by ketosis also reduces the toxic effects of excitatory pathways in our brains. Furthermore, recent data suggests that ketosis alleviates pain in addition to having an overall anti-inflammatory effect. [7]
The ketogenic diet acts on multiple levels at once, something that no drug has been able to mimic. This is because mitochondria are specifically designed to use fat for energy. When our mitochondria use fat as an energetic source, its toxic load is decreased, the expression of energy producing genes are increased, its energetic output is increased, and the load of inflammatory energetic-end-products is decreased.
The key of these miraculous healing effects relies on the fact that fat metabolism and its generation of ketone bodies (beta-hydroxybutyrate and acetoacetate) by the liver can only occur within the mitochondrion, leaving chemicals within the cell but outside the mitochondria readily available to stimulate powerful anti-inflammatory antioxidants. The status of our mitochondria is the ultimate key for optimal health and while it is true that some of us might need extra support in the form of nutritional supplementation to heal these much needed energy factories, the diet still remains the ultimate key for a proper balance.”
More here: http://www.drmyhill.co.uk//Ketogenic diet a connection
FASTING RELEASES HUMAN GROWTH HORMONE
“HGH secretion was assessed in 7 men during a control period and during starvation. Plasma samples were collected hourly for 24 hr in each period. Starvation for 72 hr increased growth hormone secretion in each individual. Overall, fasting resulted in a 2½ to 3-fold increase of growth hormone secretion. Secretion of hGH was greater in premenopausal women than in men during a control period, but no significant increase of hGH secretion occurred in women after 72 hr of fasting.” – See more at:
http://press.endocrine.org/doi/pdf/10.1210/jcem-39-2-385
TRUE KETOSIS VS FAKE KETOSIS
True ketosis comes from starvation (fasting) or severe carbohydrate restriction. There are no high glucose levels under these conditions. The ketones come from the actual break down of body fat.
False ketosis is the result of coconut oil or mct oil. These provide ketones while being able to eat massive amounts of carbohydrates. The ketones are NOT coming from body fat but the coconut or mct oil. Yes ketostix will say you are in ketosis but you are really in fake ketosis.
You want TRUE not fake ketosis. Avoid coconut or mct oil until you are in TRUE ketosis and maintain less than 30g carbs a day.
IF you are at optimal weight and not trying to lose fat then yes coconut or mct oil and low glycemic load carbohydrates is not a problem.
*For someone on a KETOGENIC diet for epileptic purposes this gives them the advantage of more carbs. For someone trying to lose fat this is a disadvantage because your body will use the coconut oil ketones over body fat ketones for energy.
ALCOHOL STOPS FAT LOSS
The quickest way to halt weight loss and put on fat is alcohol. Why? Because alcohol is the body’s first choice to burn for fuel. Guess what happens to any food you mix with it? That’s right it goes straight to your waistline until the alcohol is burned off.
AVOID alcohol until you reach ideal weight. The fastest way to get over alcohol addiction is a 72 hour dry fast— your brain is purged clean with ZERO CRAVINGS.
HERES TONS OF ARTICLES ON IT: https://www.google.com/search
SUGAR: PURE, WHITE AND DEADLY
https://ia902205.us.archive.org/…/YUDKINJohn-Pure_White
The type of food recommended should be low glycemic load. Chart here:
http://www.lowglycemicload.com/glycemic_table.html
You want minimal insulin spikes. Insulin and insulin resistance rapidly ages you. More here: https://scholar.google.com/scholar…
Vegan or non vegan isn’t as important as balanced and nutrient dense. If you are vegan be vegan if you feel best with plants and meat do that. DO EAT PLANTS regardless.
Meat should only be eaten alone or with vegetables never starch or fruit. Salad, meat fish or chicken is a perfect pairing.
If vegan be sure to choose low glycemic load vegetables, fruits, nuts seeds and grains. http://www.lowglycemicload.com/glycemic_table.html (stay as close to 15 as possible)
VEGAN KETO IDEAS:
https://www.onnit.com/academy/vegan-keto-diet/
LOW CARB VEGETARIAN IDEAS:
http://ketodietapp.com/…/81-delicious-savory-low-carb-veget…
LOW GLYCEMIC LOAD FRUITS:
http://adrenalfatiguesolution.com/fruits-lowest-glycemic-l…/
What about fruit juice fasting??
Waste of time and money; here’s why:
#1 A detox without autophagy IS NOT A DETOX. Autophagy means “to eat self” — that would be the junk weak sick cellular matter.
#2 Autophagy is induced by ketosis which is induced by fasting or extreme protein/carbohydrate restriction.
#3 A fast is NOT drinking a bunch of sugary bottled fruit juice which does NOT induce either ketosis or autophagy.
***Next person that wants to sell you a “detox kit” ask them point blank, “Does this inhibit mTOR, activate AMPK, promote autophagy or induce ketosis?”
If they look at you like a deer in headlights say, “No thanks.”***
Fructose which converts in liver to Glucose halts not only Ketosis but mTOR inhibition, AMPK activation AND autophagy (the whole point of fasting!!!) especially if you ingest more than 30g a day— thats about one 8oz glass of fruit juice. So instead of “juice feasting” all day long accomplishing nothing but wasting a bunch of money and overdosing on fructose an intelligent strategy would be to eat your fruit whole WITH IT’S FIBER (less blood sugar spikes) at the BEGINNING of your feeding window/pre workout.
Fruit without fiber is straight fructose.
Here’s what’s wrong with too much fructose:
Fructose Utilization and Associated Metabolic Dysfunction
“Consumption of fructose has been shown to be highly correlated with the development of diabetes, obesity and the metabolic syndrome. Consumption of soft drinks (high in HFCS) is associated with an increased risk for obesity in adolescents and for type 2 diabetes in young and middle-aged women. Excess fruit juice (also rich in fructose) is associated with the development of obesity in children.
Consumption of fructose by laboratory animals results in their developing several features of metabolic syndrome, including obesity, visceral fat accumulation, fatty liver, and elevated insulin and leptin levels. It is likely that the increase in leptin following fructose consumption represents leptin resistance, which could account for the increased food intake observed in fructose-fed animals.”
http://themedicalbiochemistrypage.org/fructose.php
Choose high nitric oxide producing plants like beets and arugula. More on nitric oxide and longevity here:
https://scholar.google.com/scholar…
Cancer and its relationship with metabolism, calorie restriction, mTOR inhibition, AMPK activation, autophagy and glucose:
https://scholar.google.com/scholar…
AVOID ADDING SUGAR TO ANYTHING.
************************************
LIST OF GLUCOSE DISPOSAL AGENTS
You DO NOT want your blood sugar spiking and releasing insulin. You want the highest insulin sensitivity possible. These Glucose Disposal Agents in conjunction with dry fasting can assist in regulating blood sugar.
R-ALA (in interstellar blend thermo)
R-Alpha Lipoic Acid (R-ALA), an active isomer of Lipoic Acid, is easily synthesized and metabolized by the body. R-ALA is 2 times more effective than regular (RS) Alpha Lipoic Acid and is a key component of mitochondrial dehydrogenase, which may help to slow the natural aging process in animals. Alpha lipoic acid is known as the “universal antioxidant” because of its unique solubility in both water and fat. It has the ability to conserve and prolong the life of vitamins C & E in the body, increasing their effectiveness. Alpha lipoic acid is also an excellent metal detoxifier, particularly for mercury and cadmium, which it binds to and neutralizes for excretion.
BITTER MELON (in interstellar blend thermo)
Four cucurbitane glycosides, momordicosides Q, R, S, and T, and stereochemistry-established karaviloside XI, were isolated from the vegetable bitter melon (Momordica charantia). These compounds and their aglycones exhibited a number of biologic effects beneficial to diabetes and obesity. In both L6 myotubes and 3T3-L1 adipocytes, they stimulated GLUT4 translocation to the cell membrane—an essential step for inducible glucose entry into cells. This was associated with increased activity of AMP-activated protein kinase (AMPK), a key pathway mediating glucose uptake and fatty acid oxidation. Furthermore, momordicoside(s) enhanced fatty acid oxidation and glucose disposal during glucose tolerance tests in both insulin-sensitive and insulin-resistant mice. These findings indicate that cucurbitane triterpenoids, the characteristic constituents of M. charantia, may provide leads as a class of therapeutics for diabetes and obesity.
http://ac.els-cdn.com/S1…/1-s2.0-S1074552108000823-main.pdf
Gymnema sylvestre (in interstellar blend thermo)
Gymnema sylvestre is native to India, and its leaves are a traditional folk medicine for diabetes. Several studies in humans and animals have confirmed it can improve glucose control, possibly by inhibiting glucose uptake in the small intestine and enhancing insulin release. It may even have positive effects on lipid metabolism and has potential for use in treating diabetes and obesity.
Methanolic leaf extract of Gymnema sylvestre augments glucose uptake and ameliorates insulin resistance by upregulating glucose transporter-4, peroxisome proliferator-activated receptor-gamma, adiponectin, and leptin levels in vitro.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4835989/
Berberine (in interstellar blend thermo)
AMP-activated protein kinase (AMPK) and sterol regulatory element binding protein (SREBP)-1c are major therapeutic targets in the treatment of metabolic diseases. In the present study, the fat-reducing mechanisms of berberine (BBR), a natural isoquinoline, was investigated by examining the AMPK-mediated modulation of SREBP-1c in 3T3-L1 adipocytes. BBR activated AMPK in a dose- and time-dependent manner, and increased the phosphorylation of the 125-kDa precursor form of SREBP-1c, which suppressed its proteolytic processing into the mature 68-kDa form and its subsequent nuclear translocation. The binding of nuclear SREBP-1c to its E-box motif-containing target DNA sequence was decreased following treatment with BBR, which led to a decrease in the expression of lipogenic genes and subsequently reduced intracellular fat accumulation. Transfection with AMPKα1 siRNA, and not control siRNA, inhibited BBR-induced phosphorylation of the 125-kDa SREBP-1c, which confirmed that AMPK was responsible for phosphorylating SREBP-1c.
AMPKα1 siRNA transfection rescued the proteolytic processing, nuclear translocation and target DNA binding of SREBP-1c that had been suppressed by BBR. In addition, BBR-induced suppression of lipogenic gene expression and intracellular fat accumulation were rescued by AMPKα1 siRNA transfection. In conclusion, the results of the present study demonstrate that BBR activates AMPK to induce phosphorylation of SREBP-1c, thereby suppressing proteolytic processing, nuclear translocation and target DNA binding of SREBP-1c, which leads to a reduction in lipogenic gene expression and intracellular fat accumulation. The results of the present study indicate that BBR may be a potential candidate for the development of drugs to treat obesity.
Chromium
“Chromium Enhances Insulin Responsiveness via AMPK
Trivalent chromium (Cr3+) is known to improve glucose homeostasis. Cr3+ has been shown to improve plasma membrane-based aspects of glucose transporter GLUT4 regulation and increase activity of the cellular energy sensor 5′ AMP-activated protein kinase (AMPK). However, the mechanism(s) by which Cr3+ improves insulin responsiveness and whether AMPK mediates this action is not known. In this study we tested if Cr3+ protected against physiological hyperinsulinemia-induced plasma membrane cholesterol accumulation, cortical filamentous actin (F-actin) loss and insulin resistance in L6 skeletal muscle myotubes. In addition, we performed mechanistic studies to test our hypothesis that AMPK mediates the effects of Cr3+ on GLUT4 and glucose transport regulation. Hyperinsulinemia-induced insulin-resistant L6 myotubes displayed excess membrane cholesterol and diminished cortical F-actin essential for effective glucose transport regulation. These membrane and cytoskeletal abnormalities were associated with defects in insulin-stimulated GLUT4 translocation and glucose transport. Supplementing the culture medium with pharmacologically relevant doses of Cr3+ in the picolinate form (CrPic) protected against membrane cholesterol accumulation, F-actin loss, GLUT4 dysregulation and glucose transport dysfunction. Insulin signaling was neither impaired by hyperinsulinemic conditions nor enhanced by CrPic, whereas CrPic increased AMPK signaling. Mechanistically, siRNA-mediated depletion of AMPK abolished the protective effects of CrPic against GLUT4 and glucose transport dysregulation. Together these findings suggest that the micronutrient Cr3+, via increasing AMPK activity, positively impacts skeletal muscle cell insulin sensitivity and glucose transport regulation.
Vanadyl Sulfate
Under normal conditions, the body contains 20 to 25 mg. of vanadium, and the average diet supplies about 2 mg. of vanadium per day. Food sources rich in vanadium include pepper, dill, radishes, eggs, vegetable oils, buckwheat and oats. Because of their organic environment, these natural sources are likely to be safer than over the counter preparations.
Ceylon Cinnamon (in interstellar blend thermo)
“Cinnamon Extract Enhances Glucose Uptake in 3T3-L1 Adipocytes and C2C12 Myocytes by Inducing LKB1-AMPActivated Protein Kinase Signaling
In conclusion, the current study demonstrated for the first time that CE stimulates LKB1-mediated phosphorylation of AMPK to increase glucose uptake in 3T3-L1 adipocytes and C2C12 myotubes. Since insulin resistance has been linked with the progressive development of type 2 diabetes mellitus, natural compounds that stimulate AMPK activity should have a significant clinical impact on type 2 diabetes mellitus.”
http://journals.plos.org/plosone/article/file?id=10.1371/journal.pone.0087894&type=printable
Cissus quadrangularis (in interstellar Blend thermo)
Cissus quadrangularis is also known as “Veld Grape.” It’s another medicinal plant native to Africa, India, and other parts of Asia. Cissus has traditionally been used to treat a variety of ailments such as bone fractures, ulcers, wounds, indigestion and asthma. Animal studies have shown Cissus extracts may have gastroprotective effects, contribute to bone health, and have antioxidant/antimicrobial activities. A number of people swear by Cissus as an analgesic and use it to treat weight lifting injuries. Lately, Cissus has also been touted as a fat loss agent, thanks to two studies.The first was discussed by Paul in his review of Cylaris. Of course, this study actually tested the entire Cylaris formula—which makes it difficult to draw conclusions about Cissus itself. A second study, however, did test a proprietary Cissus extract (CQR-300) and concluded it “…brought about significant reductions in weight and blood glucose levels, while decreasing serum lipids thus improving cardiovascular risk factors.”
R-ALA (in Interstellar Blend thermo)
(Na RALA is the sodium salt form of R-Lipoic Acid) Sodium R-Lipoate (Na-r-ALA)
ALA is naturally produced in the body as a mitochondrial enzyme cofactor. It is important to aerobic metabolism. ALA has been shown to increase cellular uptake of glucose to cell membranes by recruiting the glucose transporter GLUT4. ALA improves skeletal muscle glucose transport, resulting in desirable blood glucose disposal and increased muscle glycogen concentrations. Studies also indicate that in muscle, ALA is a potent anti-oxidant which protects cells from oxidative stress-induced insulin resistance. ALA is an invaluable addition that offers a multitude of benefits plus countless extras for any serious athlete.
Banaba (in Interstellar Blend thermo)
Lagerstroemia speciosa is Banaba—a tree native to Southeast Asia. The leaves have been used in traditional medicine in the Phillipines as a treatment for diabetes. As it turns out, the leaves are high in corosolic acid, which has been shown to improve glucose control in human and animal studies. It appears to do this by stimulating glucose uptake in muscle cells. Glucose transport is mediated by specific transporter proteins. Corosolic acid acts by increasing the amount of a particular transporter (GLUT-4) on the cell membrane surface. Although more work remains to be done, banaba looks like a useful ingredient for treating hyperglycemia and diabetes.
Gynostemma Pentaphyllum (in Interstellar BlendS)
“Gynostemma pentaphyllum is a plant distantly related to the cucumber. In traditional Asian medicine, it’s used to promote longevity. Today’s scientists have discovered why Asian doctors prescribed G. pentaphyllum to address age-related health issues: It promotes AMPK activation.
G. pentaphyllum not only activates AMPK, but it also shuttles excess fats into the mitochondria to be utilized for energy and safe disposal. The result is efficient energy production and a sharp reduction in unnecessary fat storage.
Results of G. pentaphyllum-induced AMPK activation include increased fat burning, as well as an increase in cellular glucose uptake. Extracts of G. pentaphyllum have other beneficial properties as well, including the ability to prolong cellular life in the face of stresses induced by oxidation, fat accumulation, and diabetes.
When scientists began exploring the benefits of G. pentaphyllum for AMPK activation, they turned to animal studies. What they found was that leaf extracts of G. pentaphyllum activate AMPK, resulting in reduced body weight gain and fat accumulation. In a preclinical study, obese mice supplemented with G. pentaphyllum showed impressive declines in markers associated with obesity and its related diseases.
In another study, this time using diabetic rats, three weeks of G. pentaphyllum supplementation resulted in improved glucose tolerance by 35% and reduced new glucose production in the liver by 29%, with a reduction in liver glycogen, the storage form of sugar.
These results show the enormous beneficial impact of reducing circulating sugar and fats in response to AMPK activation by G. pentaphyllum.
Human studies have confirmed what many of the researchers had found in the lab: G. pentaphyllum boosts AMPK activity and provides important longevity benefits.
In a compelling human study, type II diabetics who were not using diabetic medications drank a tea made with G. pentaphyllum. The results compared to controls were:
A 5-fold reduction in fasting glucose,
A 10-fold reduction in hemoglobin A1c, a measure of chronic glucose exposure,
A near 3-fold decrease in insulin resistance,
No dangerously low blood sugar episodes, which can often occur with certain oral antidiabetic drugs (especially sulfonylurea class drugs). In another human study, those taking G. pentaphyllum significantly boosted the effects of a sulfonylurea antidiabetic drug, producing an additional fasting glucose reduction of 52.2 mg/dL compared with just 16.2 mg/dL for the drug alone.
None of these findings should be surprising since the prescription drug metformin, which is an AMPK activator, produces many of these same benefits.”
http://www.lifeextension.com/magazine/2014/ss/ampk/page-01
What would I do with my current understanding if I had cancer?
New blend specifically designed to activate AMPK, SIRT1, FOXO3, and Autophagy while inhibiting, PI3K, AKT, Sonic Hedgehog, IGF1 and mTOR coming in 2018!
Well it can survive with glucose, fructose, mct and amino acids…. So what’s left?
Fat. What else is associated with that? Fasting—which leads to ketosis which leads to mTOR inhibition, AMPK activation, autophagy as well as p53, sirt1, foxo and a whole bunch of other anti cancer stuff.
I would basically dry fast and hold deep level ketosis (160) along with KETOGENIC diet until I killed it.
I would also employ longevity herbs or agents that inhibited mTOR, activated AMPK and promoted autophagy like what’s in Interstellar Blend— Gynostemma, Reishi, Ginseng, Astragalus, Rhodiola, He Shou Wu etc the drugs Metformin and Rapamycin would be of interest as well.
I would definitely NOT be eating fruit or any glucose or fructose containing foods; I would also be VERY careful on protein; in fact I might just nix all 3 until cancer aborted. Fat only diet.
OLIVE OIL inhibits mtor.
I would also use baking soda or milk of magnesia to keep my body ph alkaline while deep level ketosis.
To keep nitric oxide levels high I would employ neo40 or arugula.
https://www.neogenis.com/products/neo40-daily/
Last but not least I would watch lots of comedies, read lots of funny things and laugh as often as possible—I would keep all negativity of any form out of my mind.
FEAR
You conquer fear by facing it.
A dry fast is the conquering of fear. Fear of what? Fear of going without food, fear of going without water, without pills, without vitamins, supplements, weed, nicotine, caffeine, alcohol, drugs….whatever.
What else?
That’s right, fear of the unknown. Fear of death. Fear of losing control. Fear of breaking. Fear of being vulnerable. Fear of failing. Fear of being revealed.
To the timid this seems like insanity.
No training wheels, filters, buffers, numbing agents, masks, props or diversions— just YOU and nothing else—nowhere to run and nothing to hide behind.
The body a sealed vessel….
EXCELLENT.
THIS is how true strength of character and true strength of will is forged.
No mere mortals dry fast.
These ARE no ordinary human beings.
These are EXTRAORDINARY BEINGS.
“No water or food you say?”
Yes….
With pleasure….
BRING IT!
DANGERS OF CHRONIC DEHYDRATION
You DO NOT want to stay chronically dehydrated; some people confuse dry fasting as a way of life; like never drink water. This is idiotic. It is HORMESIS which is like a little taste of death to stress test the system and ideally eradicate disease and weakness in the process. The dose makes the poison. IF you stay in a dehydrated state you over express certain hormones and other things to try and counteract this to survive that will cause all sorts of problems like metabolic syndrome and cardiovascular issues as well as blunt and hinder some of the anti aging mechanisms in your body. When you are NOT dry fasting you should be HYDRATING— big time.
One example of this is chronic dehydration over expresses the water storing hormone aldosterone which also in the process shuts down klotho the anti aging gene; this causes a bunch of problems and premature mortality.
Downregulation of Klotho expression by dehydration
Klotho, a transmembrane protein, protease, and hormone mainly expressed in renal tissue counteracts aging. Overexpression of Klotho substantially prolongs the life span. Klotho deficiency leads to excessive formation of 1,25(OH)2D3, growth deficit, accelerated aging, and early death. Aging is frequently paralleled by dehydration, which is considered to accelerate the development of age-related disorders. The present study explored the possibility that dehydration influences Klotho expression. Klotho transcript levels were determined by RT-PCR, and Klotho protein abundance was detected by Western blotting in renal tissue from hydrated and 36-h-dehydrated mice as well as in human embryonic kidney (HEK293) cells.
Dehydration was followed by a significant decline of renal Klotho transcript levels and protein abundance, accompanied by an increase in plasma osmolarity as well as plasma ADH, aldosterone, and 1,25(OH)2D3 levels. Antidiuretic hormone (ADH; 50 nM) and aldosterone (1 μM) significantly decreased Klotho transcription and protein expression in HEK293 cells. In conclusion, the present observations disclose a powerful effect of dehydration on Klotho expression, an effect at least partially mediated by enhanced release of ADH and aldosterone.”
http://www.ncbi.nlm.nih.gov/pubmed/21734097
Dehydration: a new modulator of klothoexpression
What’s the lesson?
Use dry fasting as a tool not as a way of life keeping an individual in a state of chronic dehydration.
There is a time to dry fast and a time to be well hydrated. Intelligence is knowing when to apply each.
JOIN DRY FAST GROUP HERE:
Gavin Robert McGowen
THE INTERSTELLAR PLAN
ACTIVATE YOUR SUPERPOWERS!!
www.theinterstellarplan.com
PLEASE SHARE THIS PAGE SO OTHERS MAY LEARN HOW TO HEAL THEMSELVES AS WELL.